Hydrogen Cyanide Electron Geometry

Alright, gather 'round, folks! Let’s talk about something that sounds way more terrifying than it actually is – well, kinda. We're diving into the electron geometry of hydrogen cyanide (HCN). Don't worry, I'm not about to poison you (unless you steal my latte). Think of it more as… a molecular magic trick with a slightly sinister name.
Now, I know what you're thinking: "Cyanide? Isn't that the stuff spies use to bite down on when they're captured?" Yes, it is. But before you start picturing yourself as James Bond, let’s focus on the electrons and their funky arrangement. It's like the seating chart at a super awkward family reunion, but with, you know, deadly consequences if the chart is totally off.
The Central Character: Carbon (The Drama Queen)
Every good story needs a main character, and in our molecular drama, that's carbon. Carbon, bless its little atomic heart, wants to make friends. Like, a lot of friends. It wants to form four bonds, which is why it's always the life of the party. It's the social butterfly of the periodic table, always looking for a covalent connection.
Must Read
So, carbon is smack-dab in the middle of HCN. On one side, it’s holding hands with hydrogen (a simple, no-fuss kind of atom). On the other side, it's in a very committed relationship with nitrogen. And by committed, I mean a triple bond. Think of it as a super-strong handshake that says, "We are together forever!" Or, you know, until someone breaks the molecule apart. But let’s not get morbid.
Electron Geometry: Where the Electrons Hang Out
Electron geometry is basically a fancy way of asking, "Where are all the electrons chilling out?" It’s not just about the atoms themselves, but the regions of high electron density. These regions are formed by both bonding electrons (the ones holding atoms together) and lone pairs (the electrons that are just lounging around and not participating in bonding… the wallflowers of the atomic world).

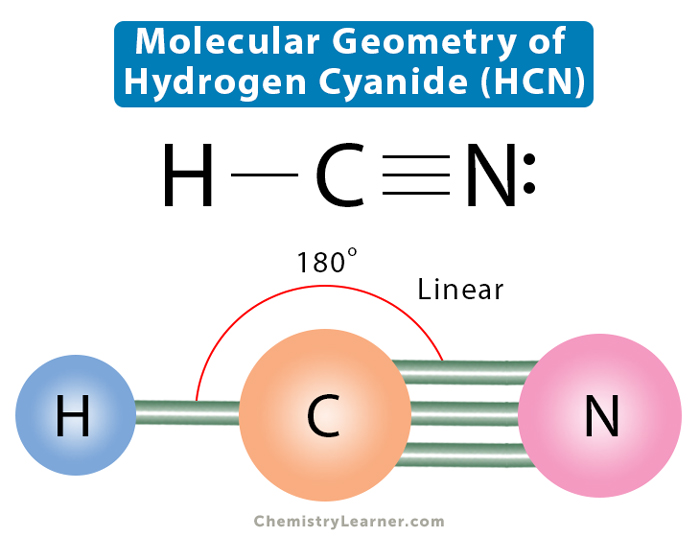
In HCN, we have two regions of high electron density around the central carbon atom. One region is the bond to hydrogen, and the other is the triple bond to nitrogen. That's it! No lone pairs, no extra baggage. Carbon is being surprisingly responsible here.
The Shape of Things: Linear, Baby!
With two regions of electron density, the electron geometry is linear. Imagine a tug-of-war. If you have one person pulling on each side, you get a straight line, right? Same principle here. The hydrogen and nitrogen are pulling the carbon in opposite directions, resulting in a 180-degree angle. It’s as straight as an arrow, or, more accurately, as straight as a line of people waiting to get into a trendy brunch spot.
Think of it like this: If HCN were a tiny, deadly barbell, carbon would be the weight in the middle, hydrogen one side and nitrogen the other. It's a perfectly balanced, albeit poisonous, exercise routine.
Molecular Geometry: Is it Different?
Now, electron geometry is related to, but not quite the same as, molecular geometry. Molecular geometry only cares about the atoms and how they're arranged, not the lone pairs. Since HCN has no lone pairs on the central atom, the electron geometry and the molecular geometry are the same: linear!
Hooray! It's like getting a two-for-one deal on geometry. We solved it once, and we're done! This is why chemistry can be fun. Sometimes.

Why Does it Matter? (Besides Bragging Rights)
Okay, so why should you care about the electron geometry of a molecule that can potentially end your existence? Well, the shape of a molecule determines its properties. This shape affects how it interacts with other molecules. For example, the linear shape of HCN makes it very polar, meaning it has a slightly positive end (hydrogen) and a slightly negative end (nitrogen). This polarity affects how it dissolves in water and how it interacts with biological molecules (like, say, the enzymes it shuts down to ruin your day.)
So, knowing the electron geometry is crucial for understanding a molecule's behavior. It's like knowing someone's personality before you decide to become their roommate. You wouldn't want to accidentally move in with a molecular Debbie Downer, would you?
In Conclusion (Before You Run Away Screaming)
Hydrogen cyanide might sound scary, but its electron geometry is surprisingly simple: linear! Carbon, with its two regions of electron density, arranges itself in a straight line between hydrogen and nitrogen. And while you might not be using this information to disarm any bombs anytime soon, you now have a slightly better understanding of the world at a molecular level. Congratulations! You've survived a chat about electron geometry. Now, who's up for another latte? I promise, no cyanide this time!

