How To Increase The Concentration Of A Solution
Ever wondered how to make your lemonade extra lemony or your coffee extra strong? The secret lies in understanding how to increase the concentration of a solution! It's a bit like magic, but it's actually just simple science. This isn't just for lab coats and beakers; understanding concentration is surprisingly useful in everyday life. Get ready to explore this fun and practical topic!
Why should you care about solution concentration? Well, for beginners, it's a foundational concept in chemistry that unlocks a whole new understanding of the world around you. For families, knowing how to adjust concentrations can be helpful in the kitchen – think perfecting that homemade vinaigrette or making sure your cleaning solutions are effective. And for hobbyists, like gardeners or DIY enthusiasts, understanding concentration is crucial for mixing fertilizers correctly or creating custom solutions for your projects.
So, what exactly does it mean to increase the concentration of a solution? Simply put, it means making the solute (the substance being dissolved, like sugar in water) more abundant relative to the solvent (the substance doing the dissolving, like the water). Think of it like this: imagine a glass of weak squash. To make it stronger, you add more squash concentrate, right? That's essentially what we're aiming to do!
Must Read
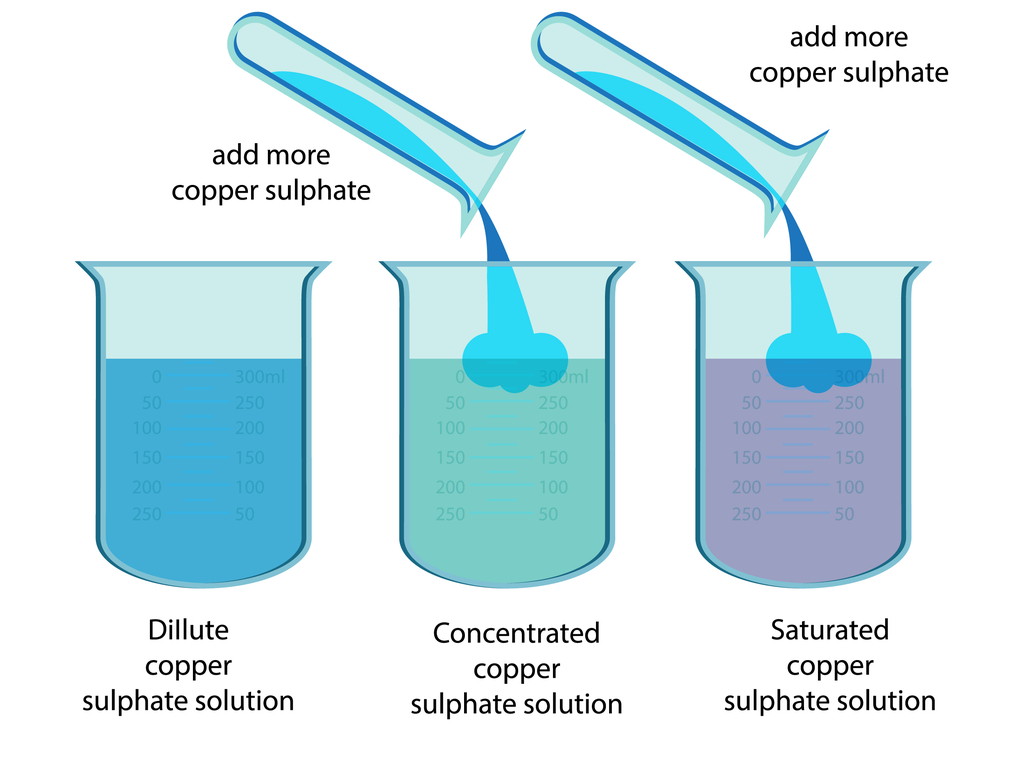
There are two main ways to increase the concentration of a solution:
- Adding More Solute: This is the most straightforward method. Just add more of the substance you're dissolving. For example, if your salt water solution isn't salty enough, add more salt. Make sure to stir well to help it dissolve completely.
- Removing Solvent: The other method involves reducing the amount of solvent. A common way to do this is through evaporation. If you gently heat your solution (like simmering tomato sauce), some of the solvent (water) will evaporate, leaving behind a higher concentration of the solute (tomato solids). Be careful not to burn the solution!
Examples and Variations: Think about making iced tea. If you brew a super strong tea concentrate and then add ice, you're diluting it slightly, but the initial high concentration means it will still be flavorful. In contrast, think about making rock candy. You start with a saturated sugar solution (a solution where no more sugar can dissolve) and then slowly evaporate the water, allowing the sugar to crystallize.

Simple Practical Tips for Getting Started:
- Start Small: When adding more solute, add it in small increments. You can always add more, but you can't easily remove it!
- Stir, Stir, Stir: Ensure the solute dissolves completely by stirring thoroughly.
- Be Patient: Evaporation takes time. Use low heat and be patient to avoid burning or splattering.
- Measure (if needed): If precision is important, use measuring spoons or cups to accurately add solute or solvent.
Understanding how to increase solution concentration isn't just about science; it's about having more control over the things you create and use every day. Whether you're perfecting your grandma's secret recipe or concocting a new fertilizer blend for your prized roses, the knowledge you've gained is sure to add an extra layer of enjoyment and success to your endeavors. So go ahead, experiment and have fun with it!



