How To Find Van't Hoff Factor

Alright, globetrotters and knowledge nomads! Ever wondered why that iced latte hits differently than you expected? Or why your homemade lemonade tastes surprisingly sour even with less sugar? The answer, my friends, might just lie in the fascinating world of the Van't Hoff factor. Don't let the name intimidate you; it's not as scary as it sounds. Think of it as the secret agent influencing how things behave in solution.
Decoding the Van't Hoff Factor: A Simple Mission
So, what exactly is this Van't Hoff factor? Simply put, it's a measure of how many particles a solute (the thing being dissolved) breaks down into when it dissolves in a solvent (usually water). Imagine dropping a sugar cube into your tea. Sugar (sucrose) dissolves, but each molecule stays intact. The Van't Hoff factor for sugar is essentially 1. But, if you drop in table salt (sodium chloride), things get more interesting! It dissociates into Na+ and Cl- ions, effectively doubling the number of particles. Its Van't Hoff factor is close to 2.
Think of it like this: you're throwing a party. Sugar is like a single friend showing up. Salt is like a couple arriving together. More guests, more impact, right? Same principle applies to solutions!
Must Read
The Formula for Fun (and Finding 'i')
Ready to crack the code? The Van't Hoff factor is usually represented by the letter 'i'. While there are fancy experimental ways to determine it precisely, we can often estimate it based on the compound's chemical formula.
Here’s the basic idea:
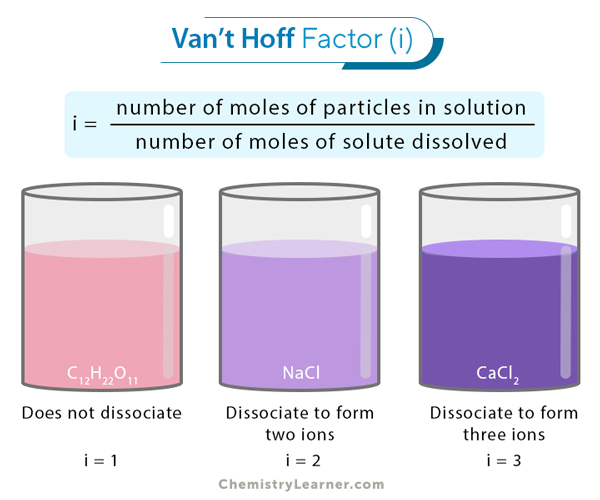
For non-electrolytes (like sugar): i = 1 (they don't dissociate).
For strong electrolytes (like NaCl, KCl): i ≈ the number of ions formed when the compound dissolves. So, NaCl -> Na+ + Cl-, therefore i ≈ 2. Similarly, CaCl2 -> Ca2+ + 2Cl-, therefore i ≈ 3.
Important Caveat: In reality, the actual Van't Hoff factor might be slightly less than the ideal value, especially at higher concentrations. This is because ions can sometimes clump together, a phenomenon called ion pairing. It's like some guests at the party deciding to stick close to their friends instead of mingling.

Putting It Into Practice: Real-World Relevance
Okay, so you know what it is and how to (roughly) find it. But why should you care? Well, the Van't Hoff factor plays a crucial role in several important properties of solutions, known as colligative properties. These properties depend on the number of solute particles, not their identity.
Here are a few examples:

- Freezing point depression: Adding salt to icy roads lowers the freezing point of water, preventing ice from forming. This is why your road trip playlist is safe!
- Boiling point elevation: Adding salt to water when cooking pasta makes the water boil at a slightly higher temperature, potentially cooking your pasta faster. (Debatable, but fun to think about!)
- Osmotic pressure: This is important in biological systems, like how cells maintain their shape. Think of it as the bouncer at the party, controlling the flow of people (water) in and out.
Tips and Tricks for the Van't Hoff Victor
Here are some quick tips to help you master the Van't Hoff factor:
- Know your electrolytes: Strong electrolytes dissociate completely, weak electrolytes dissociate partially, and non-electrolytes don't dissociate.
- Look at the chemical formula: It tells you how many ions a compound can form.
- Don't forget the 'approximately equal to' sign (≈): Real-world values might deviate slightly.
- Think of the party analogy: More particles = greater impact on colligative properties.
Think of aqueous solutions everywhere. Ocean water, beverages like gatorade, even your blood.
A Parting Thought: The Subtle Influences
The Van't Hoff factor, seemingly a small detail in the grand scheme of chemistry, reminds us that even subtle influences can have significant consequences. It's like the butterfly effect in action – a single dissolved particle contributing to a cascade of effects. So, the next time you're sipping your perfectly sweetened iced tea or salting your pasta water, remember the Van't Hoff factor and the hidden world of interactions it represents. It's a reminder that even in the simplest of things, there's always more than meets the eye.