How Many Protons Does Tin Have
You know Tin, right? The stuff in cans, sometimes in solder, maybe even those charming little toy soldiers your grandpa talks about. It's pretty chill. Keeps your beans safe. Looks a bit grey. Doesn't make a fuss. Or does it?
Most of us don't spend our days pondering the deep, dark secrets of everyday metals. We just expect them to, well, be there. Do their job. But have you ever really thought about Tin? Like, deeply? We're talking atomic level. Specifically, how many little zingy bits, those atomic VIPs called protons, does it actually have?
It's a question few dare to ask. A mystery that keeps exactly zero people up at night. And yet, it's strangely important. Because without the right number of protons, an element isn't that element. It's something else entirely. It's like trying to call your dog "Spot" when it's clearly a cat named "Mittens." Just doesn't work.
Must Read
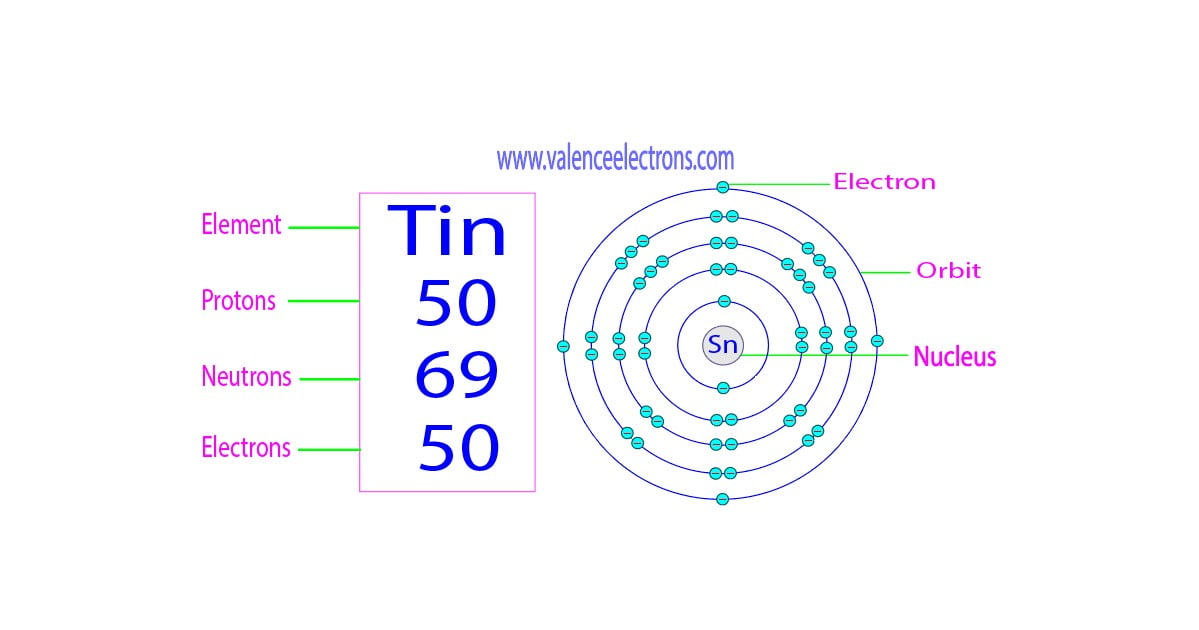
So, drumroll please! Or maybe just a polite tap on the table. Every single atom of Tin, without exception, proudly boasts exactly fifty of those glorious little protons. Not 49, not 51. Fifty. That's its ID card, its atomic fingerprint. That's what makes a thing Tin and not, say, Indium (which has 49) or Antimony (which has 51). So, to answer the initial question: Tin has 50 protons. Simple, right?
And that, my friends, is where my unpopular opinion kicks in. Because while the proton count for Tin is utterly solid, Tin itself? Oh, Tin is a bit of a diva when it comes to its full identity. It’s not just about the protons, you see. Atoms also have other residents in their tiny, bustling nuclei: the neutrons. These are the protons' chill, neutral buddies.

Here's the Controversial Bit: Tin is Just Showing Off.
Now, the number of protons defines the element. Always 50 for Tin. But the number of neutrons can change. When that happens, we get different versions of the same element called isotopes. Most elements have a few stable ones, maybe two or three that are common. Like Carbon, bless its stable heart, mostly has just two main stable isotopes. Simple. Predictable. Gets the job done.
But Tin? Oh, Tin laughs in the face of simplicity. Tin, our unassuming little grey friend, boasts a whopping ten stable isotopes! Yes, you heard that right. T-E-N. Ten different versions of itself, all with 50 protons, but varying numbers of neutrons, from 62 all the way up to 74! It's like having ten slightly different identical twins, each with a different favorite flavor of ice cream, but all sharing the same family name.

Seriously, Tin? Ten stable isotopes? Who needs that many choices? It's like asking for a coffee and being offered ten different shades of "just regular black." It's impressive, yes, but also a little bit exhausting. It's like Tin just can't make up its mind. Or maybe it just enjoys being exceptionally, ridiculously versatile. While other elements are content with a couple of identity cards, Tin carries a whole wallet full, just in case. "Oh, you need an atom with 66 neutrons today? I've got one right here! How about 70? Yup, got that too!"
It makes Tin the element with the most stable isotopes of any element on the periodic table. THE MOST! For a metal that just sits there, looking all understated in your pantry, that's quite the flex. It's an atomic overachiever, a quiet show-off. Other elements are just trying to get by, but Tin is out there collecting isotopes like they're trading cards.

So, the next time you see a humble Tin can, take a moment. Appreciate its unwavering fifty protons, yes. But also, ponder its audacious, multi-faceted nature. Its ten stable isotopes. Its refusal to conform to mere elemental simplicity. My unpopular opinion?
Tin is just doing way too much. It needs to chill out and pick a few less isotopes. But hey, maybe that's why we secretly love its overachieving, understated drama. What a show-off!

