How Many Energy Levels Does Sulfur Have

Sulfur. Yeah, that yellow stuff. Maybe you've seen it near volcanoes, or even smelled it (apologies if that memory isn't pleasant!). But did you ever stop to think about its inner life? Specifically, how many energy levels those tiny sulfur atoms are rocking?
Sulfur's Electronic Dance Party
Okay, so "energy levels" might sound a bit technical. Think of them like floors in an apartment building, but for electrons. Electrons, those negatively charged particles zipping around the nucleus, can only live on specific floors. They can't hang out in between. Each floor represents a certain energy level. The higher the floor, the more energy the electron has.
Now, sulfur. Sulfur is element number 16 on the periodic table. That means it has 16 protons and, importantly, 16 electrons when it's neutral. These electrons don't just pile on the first floor. They follow some pretty strict rules about where they can live. It’s like a cosmic bouncer is making sure only the right number of electrons are allowed on each level. Sounds intense, right?
Must Read
So, how many "floors" does sulfur need to house all 16 of its electron tenants? This is where the fun begins! It's all about filling those electron shells.
Electrons fill the energy levels closest to the nucleus first. Think of it as prime real estate! The first energy level can only hold two electrons. That's it! So sulfur uses those first two electrons to fill that floor.
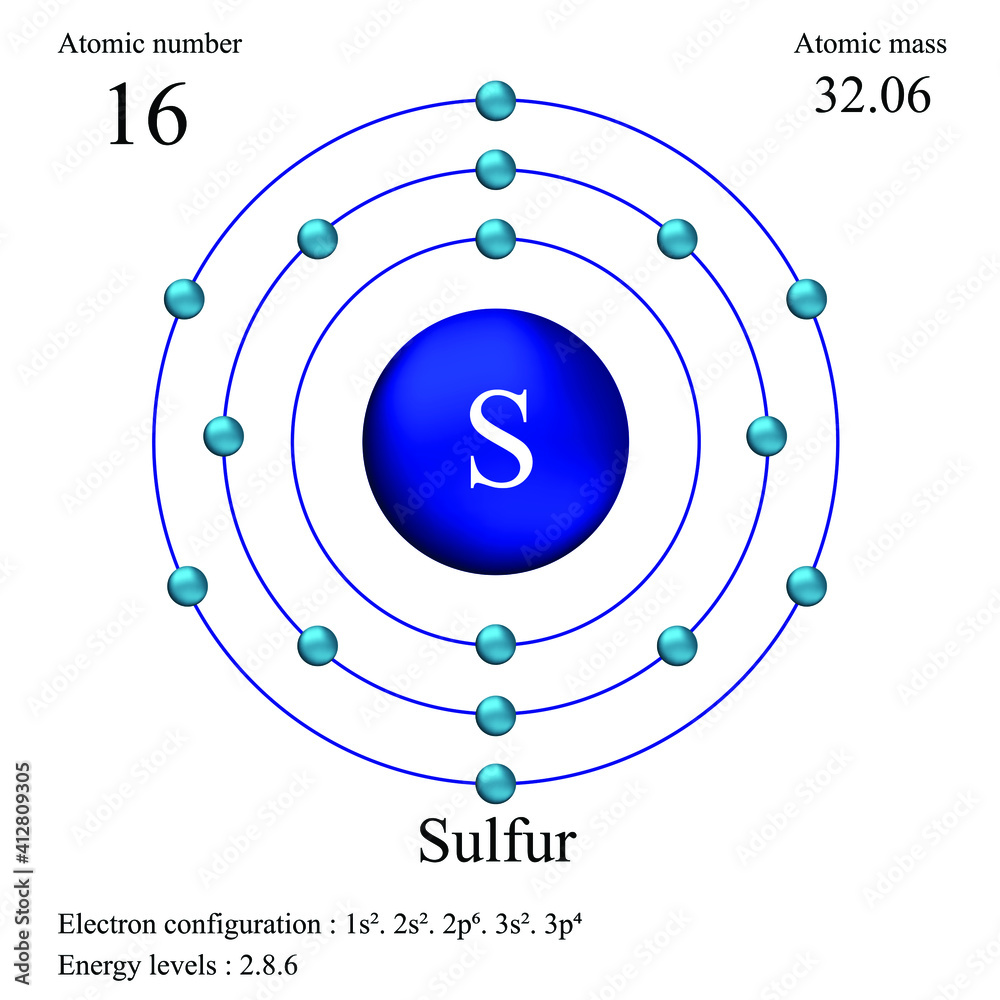
The second energy level is a little bigger and can hold up to eight electrons. Sulfur gladly plops eight more electrons there, bringing the total to ten. We're still six electrons short!
This brings us to the third energy level. This one is a bit more complicated, but for simplicity, we can say it can hold up to eight electrons as well. Because sulfur only needs six more electrons to reach its full complement of 16, these last six electrons will go to the third floor. Sulfur has its very own three-story electron apartment!

Three's Company (of Energy Levels)
So, the answer is: Sulfur has three main energy levels or electron shells that its electrons occupy. That’s not bad, right? Think of it: three distinct neighborhoods for those electrons to cruise around in.
But wait, there's more! Inside these main energy levels, things get even more interesting! Each level contains sublevels or orbitals. These sublevels are like different rooms within each floor. Some rooms are shaped like spheres (s orbitals), some like dumbbells (p orbitals), and others are even more exotic (d and f orbitals, if you're feeling adventurous!).
The arrangement of electrons in these orbitals is called the electron configuration. For sulfur, the electron configuration is 1s²2s²2p⁶3s²3p⁴. Don't freak out! It simply shows how many electrons are in each orbital. The "1s²" means two electrons in the 1s orbital, "2s²" means two electrons in the 2s orbital, and so on.

Why Should You Care?
Okay, you might be thinking: "This is cool and all, but why is it important?" Well, the number and arrangement of electrons determine how an atom interacts with other atoms. It's what makes sulfur so reactive and allows it to form all sorts of interesting compounds. Knowing how many energy levels sulfur possesses helps us understand its chemical properties.
The ability to form bonds with other atoms is dependent on its electron configuration. And, just for the record, this affects pretty much everything. From the batteries in your phone to the rubber in your tires, sulfur plays a crucial role.

So, the next time you see a volcano steaming, or even just light a match (carefully, of course!), remember those 16 electrons buzzing around the sulfur atoms, hanging out on their three energy levels. They're working hard to make the world a more interesting (and sometimes smelly) place!
Think of it this way: the number of energy levels and the electron configuration basically define sulfur's personality. It's what makes it uniquely sulfur!
Maybe understanding electron energy levels doesn't sound like your idea of a wild time. But take it from us – once you start thinking about the invisible world buzzing inside every atom, you'll realize it's way more exciting than it sounds. So go forth and explore the amazing world of chemistry. You might be surprised by what you discover! And remember, it all starts with understanding those energy levels!
