How Many Atoms Are In Copper
The Great Copper Conundrum (and why it's perfectly okay not to know)
Let's talk about copper. It's everywhere, isn't it?
From the pennies jingling in your pocket to the intricate wires humming inside your walls, copper is a true workhorse.
It’s shiny, it’s useful, and it's been around for ages. But here’s a question that might keep you up at night, or at least make you ponder for a second:
Must Read
Just how many atoms are in a piece of copper?
Now, your initial thought might be to reach for a textbook or a very smart friend. Don't bother.
Because my "unpopular" opinion on this, the one I want you to embrace, is delightfully simple:
The answer is: A ridiculous, mind-bogglingly, incomprehensibly HUGE number. And that’s all you really need to know.
Seriously. Trying to pin down an exact figure for everyday appreciation is like trying to count every single giggle at a kindergarten party.
It's not just impractical; it misses the point. The point is the joy, not the exact tally of chuckles.
Tiny Dancers: What Even Is an Atom?
So, what exactly are we talking about when we say atom?
Think of atoms as the universe's ultimate building blocks. They are unbelievably, impossibly small.
Imagine taking something tiny, like a grain of sand, and then shrinking it down until it's invisible, then shrinking it again a billion more times.
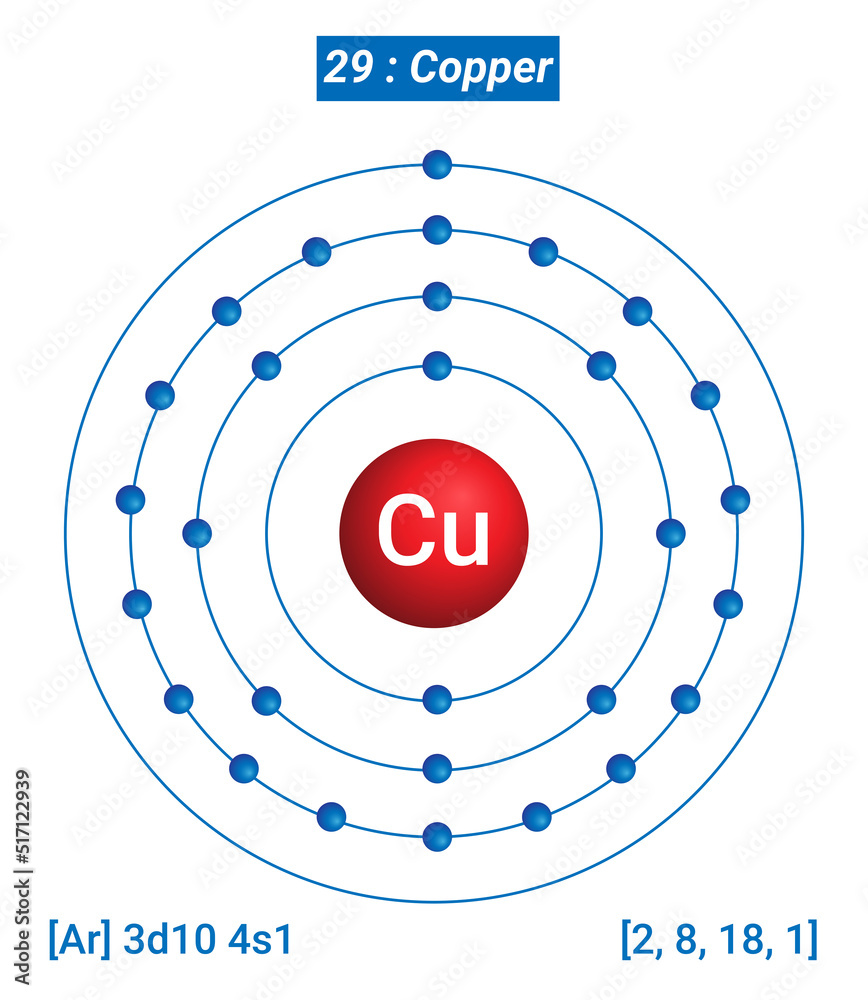
That’s the scale we're playing with here. Each piece of copper, no matter how small, is made up of these fundamental, individual copper atoms.
They are the smallest unit that still behaves like copper.
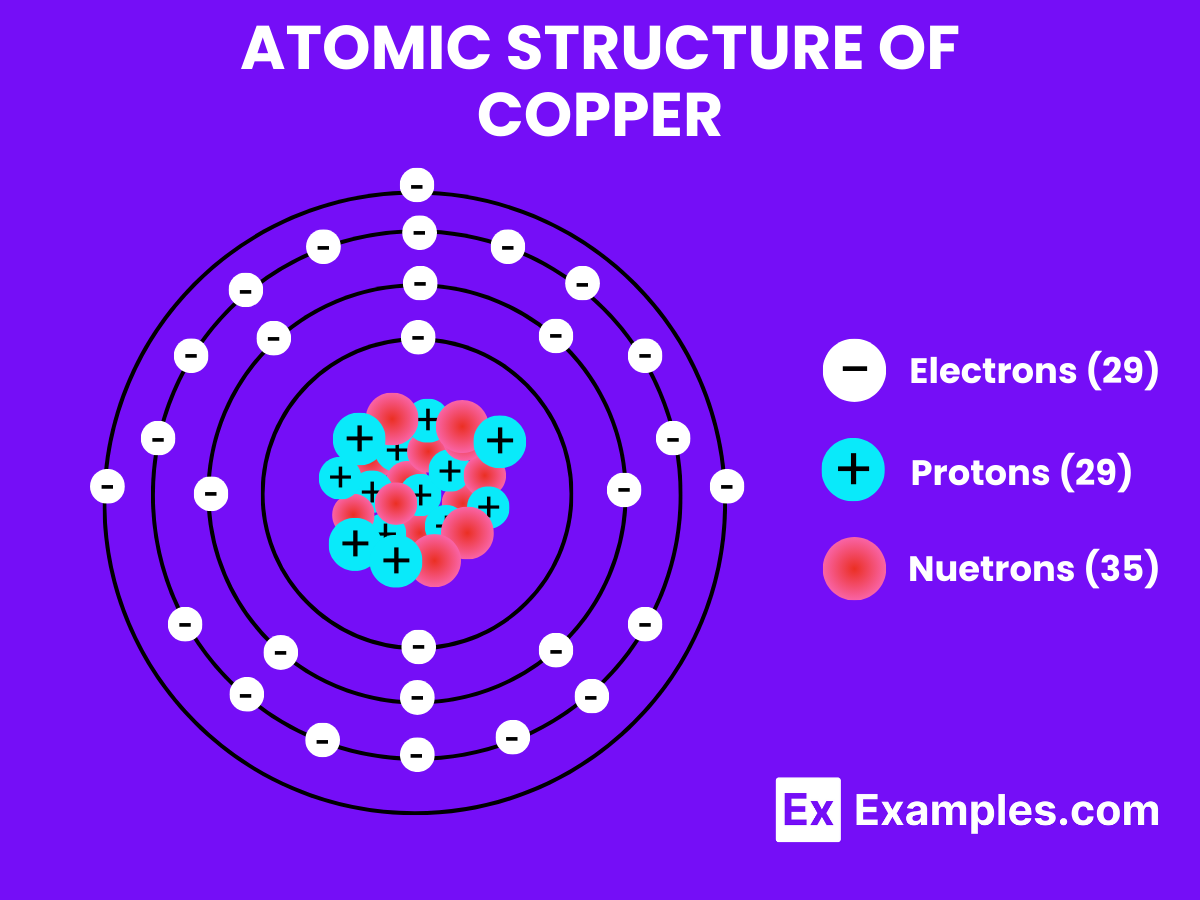
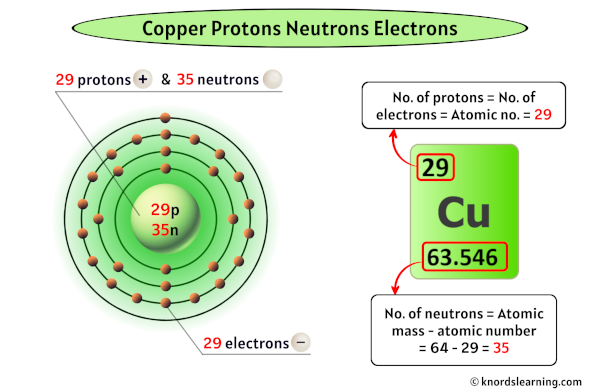
If you broke them down further, you'd get protons, neutrons, and electrons, but then it wouldn't be copper anymore. It would just be subatomic particles, which is a whole other, equally mind-bending, story.
These tiny copper atoms are constantly vibrating, wiggling, and interacting with each other.
They’re not static little marbles; they’re more like a bustling, microscopic city, always in motion.
And every single one of them contributes to the overall nature of the piece of copper you're holding.
The Numbers Game (or why we should politely decline to play)
Okay, so we know atoms are tiny. Really, really tiny. Like, "you could fit a trillion on the head of a pin and still have room for a party" tiny.
But does that help us with the "how many" question? Not really, because even if they are infinitesimally small, any visible piece of copper has an almost unfathomable quantity of them.
Scientists, bless their meticulous hearts, do have ways of estimating these numbers.
:max_bytes(150000):strip_icc()/Copper-58b602153df78cdcd83d352e.jpg)
They use very clever tricks and incredibly large numbers, like Avogadro's number (don't worry about remembering it).
It’s a number so huge that trying to write it out would fill several lines of text. It's six hundred thousand billion billion, roughly.
That's 6 with 23 zeros after it. Try saying that out loud.
It's a number that truly makes your brain feel like it's trying to fold in on itself.
Imagine counting every single drop of water in all the world's oceans. Then multiply that by a thousand. You're still nowhere near the number of atoms in even a small piece of copper.
It's the kind of number that exists more as a concept than something you could ever practically visualize or count.
And that's precisely why our "unpopular" opinion comes in handy.
Who needs the exact figure when the general idea of "an astronomical amount" is already so impressive?
Copper's Charm: More Than Just a Count
What truly makes copper fascinating isn't the precise number of atoms it contains.
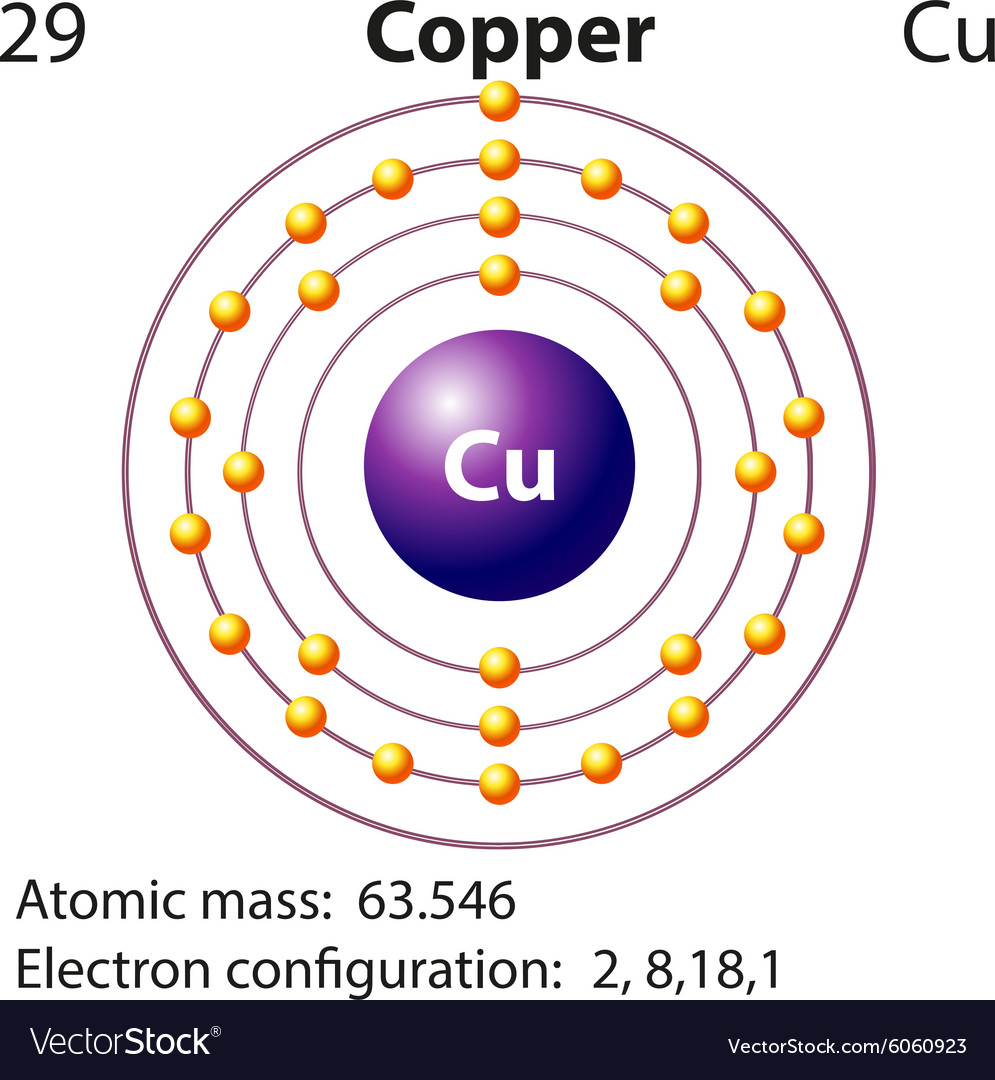
It's how those countless atoms work together to give copper its amazing properties.
Each copper atom has a unique electron configuration, especially its outermost electrons.
These electrons are a bit like free spirits, not tightly bound to any single atom, but rather able to roam freely throughout the entire piece of metal.
This freedom is what makes copper such a fantastic conductor of electricity and heat.
Those flowing electrons are the current; those vibrating atoms passing energy are the heat.
It's also why copper is so ductile and malleable – you can stretch it into thin wires or hammer it into intricate shapes without it breaking.
The atoms can slide past each other without losing their connection, maintaining the overall structure.
Think of it: billions upon billions of these tiny atomic dancers, all moving in perfect, flexible harmony, creating a material that powers our world.
That’s far more interesting and useful to ponder than an exact, unpronounceable digit.
Embracing the "Many": Our Unpopular, Yet Sensible, Opinion
So, the next time someone asks you, "How many atoms are in copper?" don't panic.
Don't try to remember some impossibly long number. Just offer them our truly enlightened, and frankly, more practical, answer.
There are SO. MANY. ATOMS. in copper that it defies easy comprehension.
And that, my friends, is enough. More than enough, actually.
It’s a beautiful thought, isn’t it? To know that every little bit of copper, every wire, every shiny coin, is a universe unto itself, teeming with these microscopic building blocks.
It’s okay to be awestruck by the sheer scale of the universe, even when it’s condensed into a penny in your palm, without needing to quantify every single speck.
Let's leave the precise counting to the super-smart scientists with their fancy equipment and endless patience.
For the rest of us, simply appreciating the vastness, the complexity, and the incredible usefulness of copper is truly a joy.
So, smile, nod knowingly, and agree with me that sometimes, "a lot" is the best, most entertaining, and utterly correct answer.
