How Is Nuclear Energy Extracted From The Earth
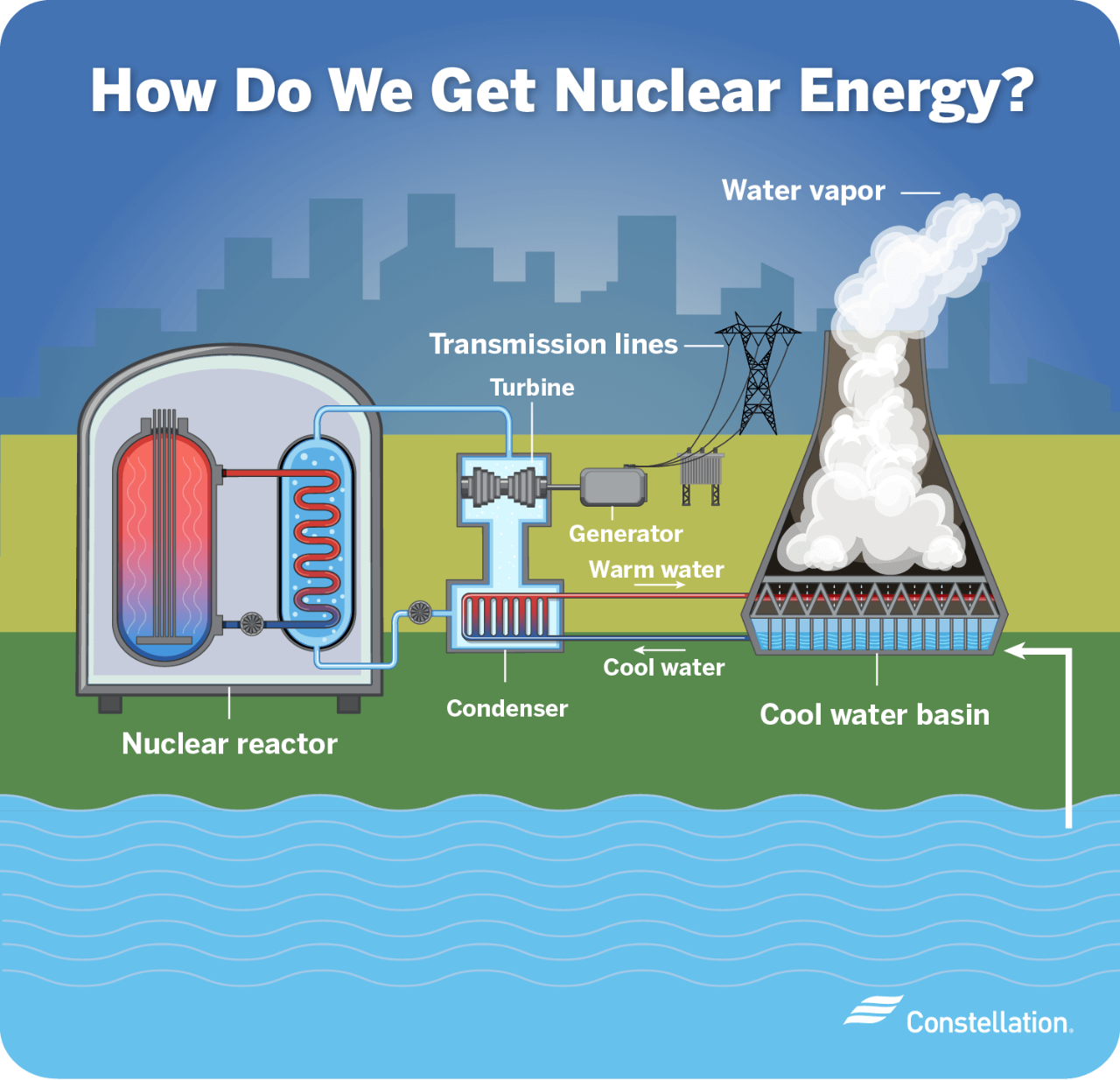
Okay, so picture this: I'm at a geology museum, staring at this chunk of rock. It's kinda dull, honestly. Looks like something you'd trip over on a hike. But the little placard next to it says "Uraninite – Highly Radioactive!" And I'm thinking, "Whoa. That ugly rock is secretly a powerhouse?" That's kind of how nuclear energy works, right? Taking something seemingly ordinary and unlocking its crazy potential.
So, how do we get that energy out of the ground? It's not like drilling for oil, folks. Time to dive into the surprisingly fascinating world of uranium mining and processing!
First things first: Finding the Stuff.
Must Read
Uranium, the rockstar of nuclear energy, isn't exactly lying around on the surface, waving hello. (Wouldn't that be convenient... and terrifying?). It's found in various rock formations, often mixed with other minerals. So, geologists, bless their clever souls, go out and explore, looking for telltale signs like uranium-rich deposits. They use things like airborne radiometric surveys (fancy!), analyzing soil and water samples, and drilling core samples. Imagine the geo-version of treasure hunting!
Once they think they've found a good spot, it's time to... you guessed it... mine it.
Mining Methods: From Open Pit to In-Situ Leaching
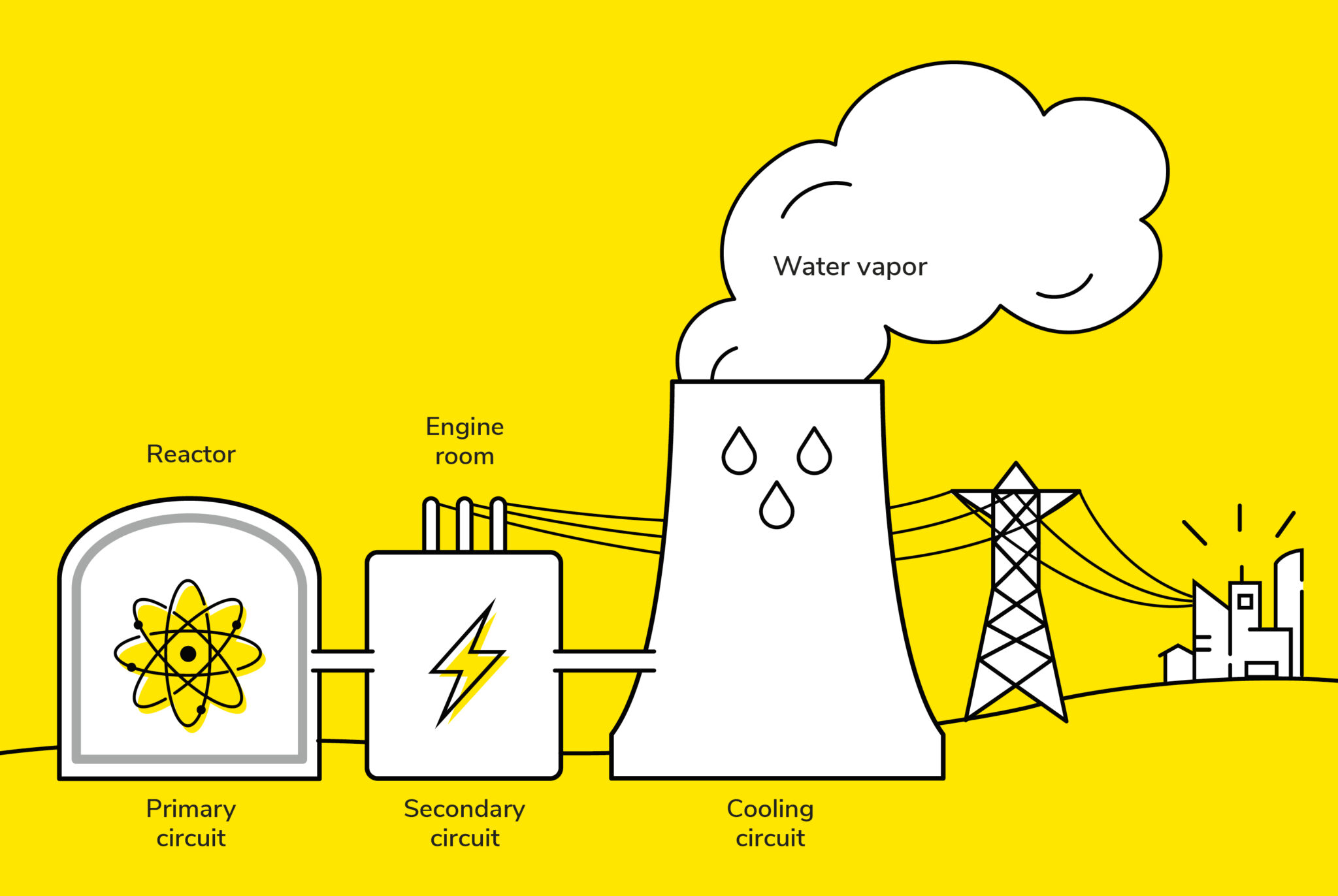
There are several ways to extract uranium ore, depending on how deep it is and the geological context. It's kinda like choosing the right tool for the job.
- Open-pit mining: Think giant, gaping holes in the earth. If the uranium ore is close to the surface, they just dig it out. It’s visually impressive (and environmentally impactful – we'll get to that later, promise!).
- Underground mining: For deeper deposits, they build tunnels and shafts to reach the ore. Picture dwarves with pickaxes (though, obviously, it’s much more modern and mechanized than that!). This method is more expensive and complex.
- In-situ leaching (ISL): Now this is interesting! They pump a solution (usually a mix of water, oxygen, and sometimes chemicals) into the uranium deposit to dissolve the uranium. Then, they pump the solution back up, now rich in uranium. It's like giving the uranium a bath and then collecting the bathwater. Seems less intrusive than digging up the whole place, right?
Side note: whichever method is used, safety is a HUGE deal. Uranium is radioactive, after all. Miners wear protective gear, and there are strict regulations to minimize exposure.
From Ore to Yellowcake: The Milling Process
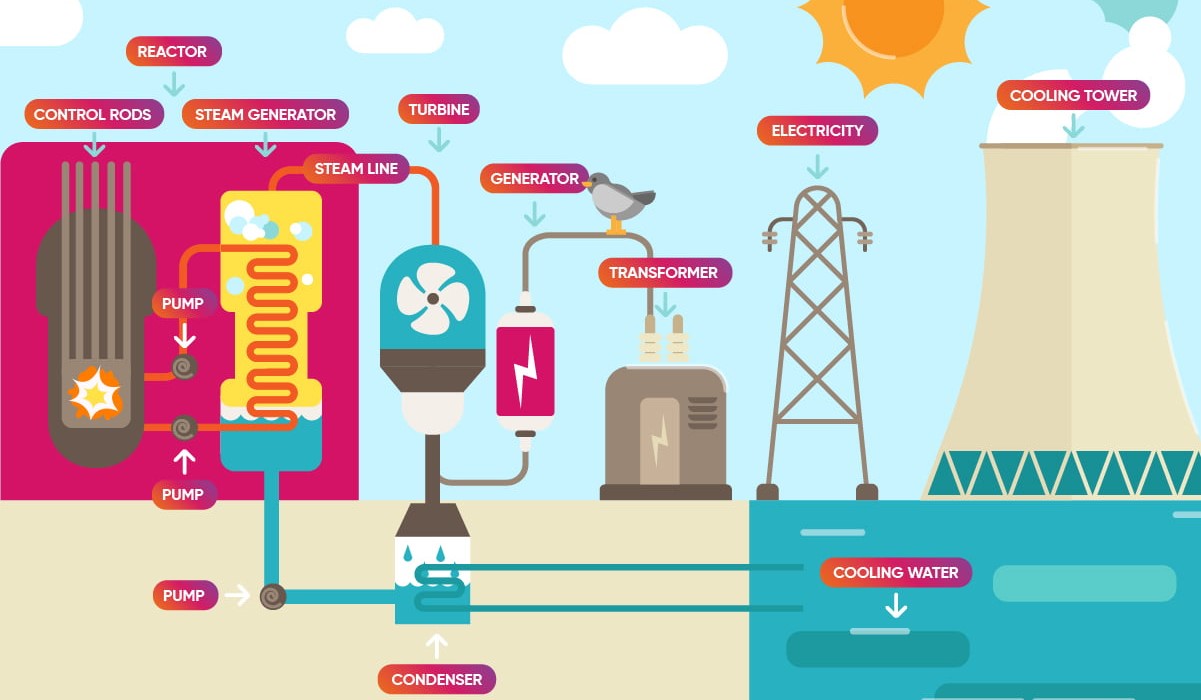
Okay, so we've got our ore. It's not ready to go into a nuclear reactor just yet! It needs to be processed. This is where the milling process comes in. The ore is crushed and ground into a fine powder. Then, it's leached (again!) with chemicals to dissolve the uranium compounds. This solution is then further processed to precipitate out a uranium concentrate.
And guess what this concentrate looks like? It's a bright yellow powder. Yep, that's why it's called "yellowcake." Kinda makes you want to bake something, doesn't it? (Don't! Seriously, don't.)
This yellowcake is primarily uranium oxide (U3O8). It's still not quite reactor-ready. Think of it as a really rough draft that needs a lot of editing.
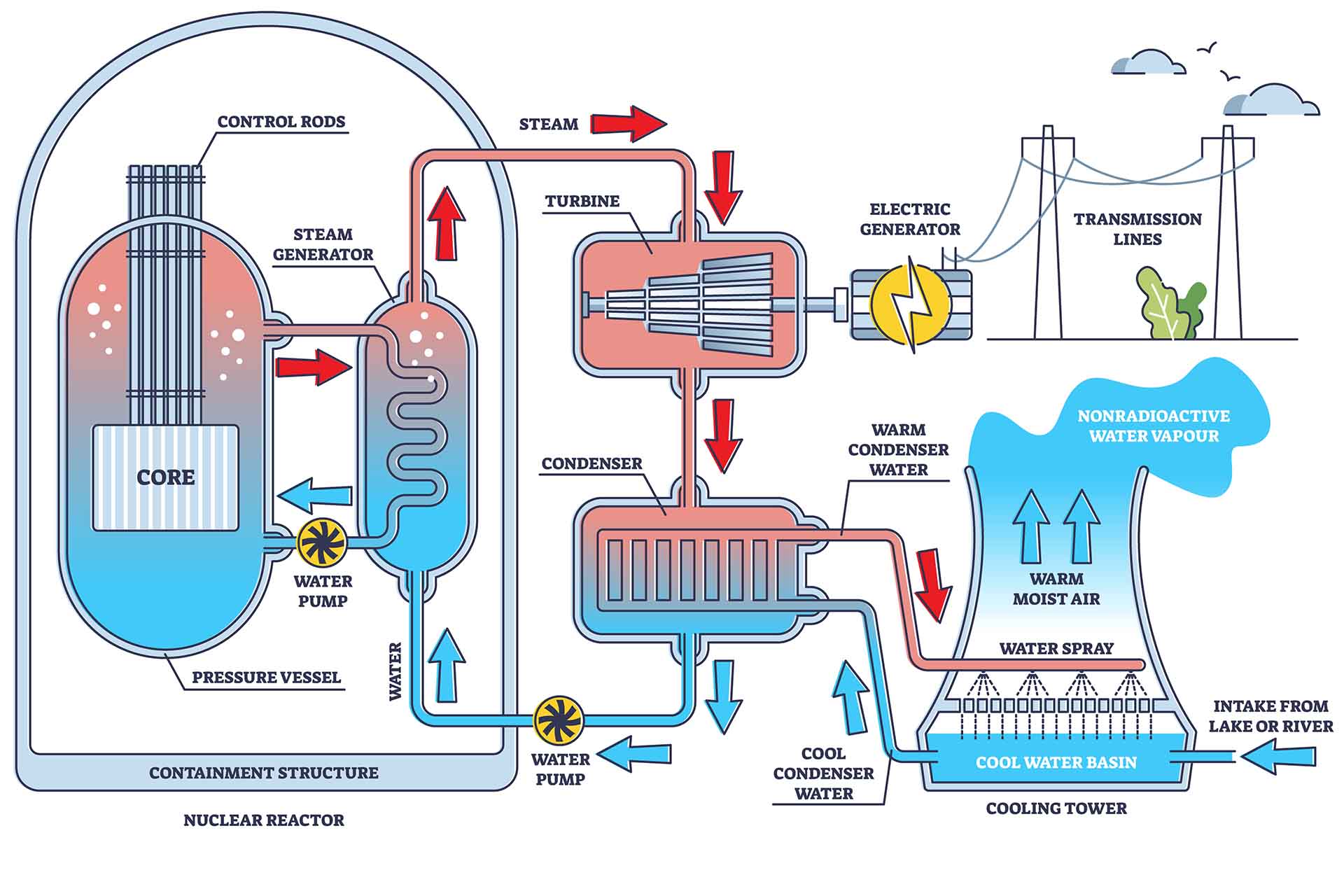
Enrichment: Making it Reactor-Worthy
Natural uranium contains two main isotopes: uranium-238 (U-238), which makes up the vast majority, and uranium-235 (U-235), which is the one that's easily fissionable (meaning it can be split to release energy in a nuclear reactor). The problem? Natural uranium only contains about 0.7% U-235. Most nuclear reactors need uranium with a much higher concentration of U-235, typically around 3-5%.
So, we need to enrich the uranium – increase the proportion of U-235. This is done using sophisticated techniques like gaseous diffusion or gas centrifuge enrichment. These methods separate the slightly heavier U-238 from the lighter U-235. It's a complex and energy-intensive process. In fact, this enrichment step is often the most technically challenging part of the entire fuel cycle.

Fuel Fabrication: The Final Touch
Finally, the enriched uranium is converted into uranium dioxide (UO2) powder, which is then pressed into small ceramic pellets. These pellets are loaded into long metal tubes (usually made of zirconium alloy) to form fuel rods. These fuel rods are then bundled together to form fuel assemblies, which are then loaded into the nuclear reactor core. And that, my friends, is how we get from a dull rock in the ground to the power source for our homes (or at least, a portion of it!).
It's a long, complicated, and fascinating journey, right? From geological exploration to high-tech enrichment, there's a lot that goes into extracting and preparing uranium for nuclear energy. And remember that environmental impact I promised? We'll have to tackle that another day, but trust me, it's an important part of the story too. Now, if you'll excuse me, I'm off to find another museum exhibit to geek out over!
