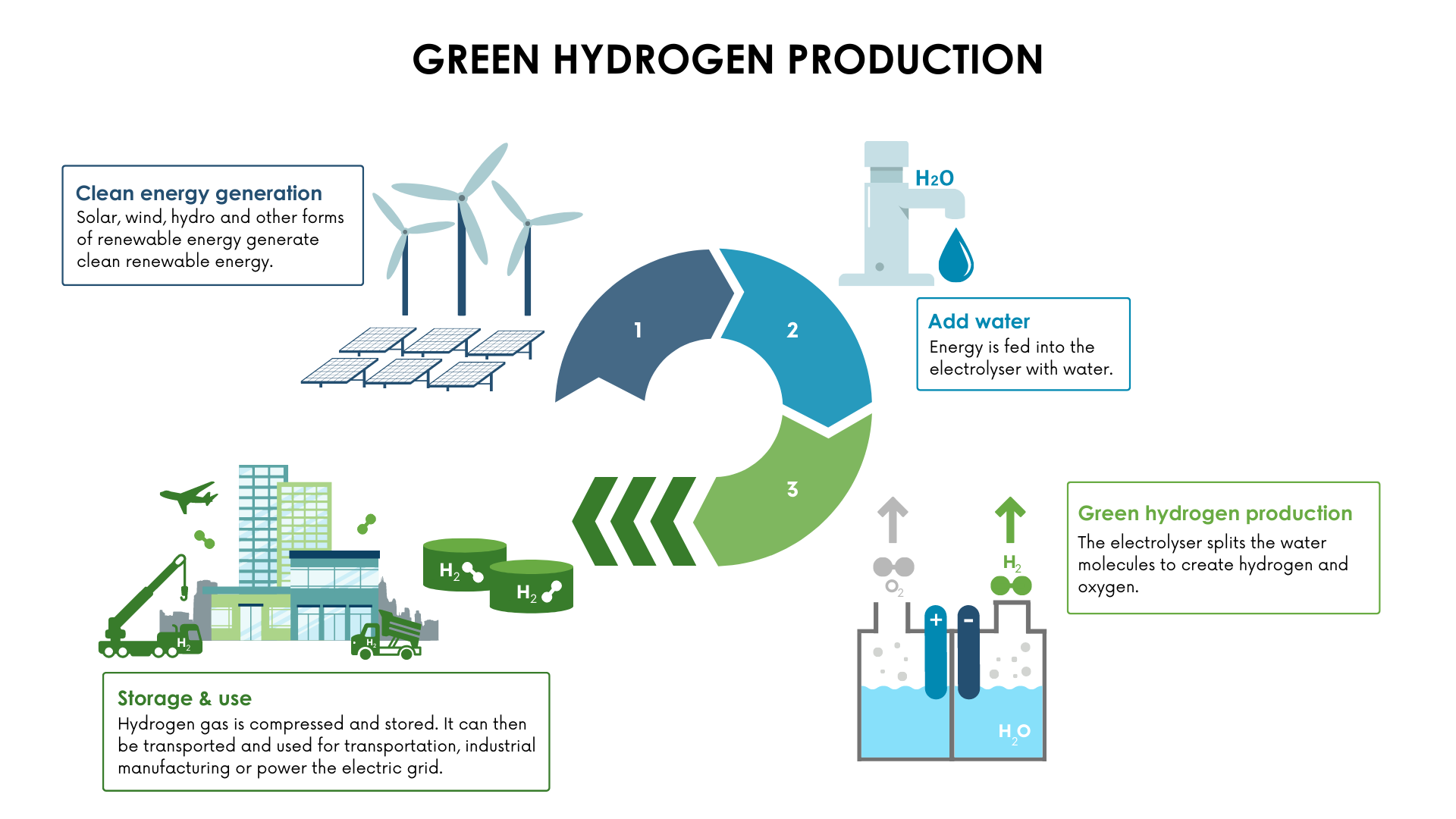
How Is Hydrogen Made From Water

Ever looked at a glass of water and wondered if it held a secret? Not just for quenching thirst or splashing around, but something truly spectacular, something that could power our world? Well, dear reader, get ready for a delightful dive into the watery wonderland where we unlock the hidden energy of plain old H2O!
Imagine water, not just as a liquid, but as a tiny, tightly-knit family of atoms. Inside every single drop, two enthusiastic Hydrogen atoms are holding hands with one dependable Oxygen atom. They’re super glued together, a perfectly happy little trio!
Our mission, should we choose to accept it, is to gently persuade these atomic buddies to let go of each other’s hands. We want to separate the lively Hydrogen from its steadfast friend Oxygen. It’s like a friendly atomic divorce, all for a good cause!
Must Read
So, how do we perform this magical separation? We introduce a little spark, a friendly nudge, in the form of electricity! Think of electricity as a super-energetic matchmaker, or perhaps a tiny, invisible hand giving these molecular friends a polite push apart.
This fantastic process, which sounds incredibly fancy but is actually quite simple, is called electrolysis. Don't let the big word scare you; it just means "splitting with electricity." It's like a secret handshake between science and nature!
The Super Simple Setup: Our Water-Splitting Toolkit
To get this atomic separation party started, we need a few simple ingredients and tools. First, of course, is our star player: water. Just plain old water will do, though sometimes we give it a tiny boost.
Next, we need a source of electricity. This can be from your wall socket, but the really exciting part is when this electricity comes from happy sunshine or gusty winds! We’ll get to that cosmic high-five in a bit.

Finally, we introduce two special metal sticks, or plates, called electrodes. Think of them as tiny, friendly magnetic poles. We dip these two electrodes right into our water.
One electrode is called the anode, and it’s like the outgoing party planner. The other is the cathode, a bit more reserved, but equally important. They're both super keen on attracting their specific atomic guests!
The Atomic Dance Party: What Happens Next?
Now, here’s where the real fun begins! When we switch on the electricity, something truly amazing happens inside that glass of water. It's like turning on the disco lights for our tiny atoms!
The electricity starts flowing, creating a tiny, invisible current through the water. This current is like a gentle but firm nudger for our H2O molecules, encouraging them to reconsider their close bond.
At the cathode, our positive electrode, the two little Hydrogen atoms from each water molecule get super excited. They see their chance for freedom! They rush over to this electrode, eager to break away from their Oxygen buddy.
"It’s like carefully separating LEGO bricks – the red ones go here, the blue ones go there, and suddenly you have two distinct piles!"
They let go of Oxygen's hand and transform into tiny bubbles of hydrogen gas! Pop! Pop! Pop! You can actually see these microscopic bubbles fizzing away, rising triumphantly to the surface.
Meanwhile, at the anode, our negative electrode, the Oxygen atoms, feeling a little left out but still resilient, gather together. They too let go and form bubbles of oxygen gas.
So, what you end up with is two separate streams of gas: pure hydrogen happily bubbling up from one electrode, and pure oxygen happily bubbling up from the other! No smoke, no fuss, just pure separation and a whole lot of potential.
A Little Boost: The "Secret Sauce"
Sometimes, plain water isn't quite enthusiastic enough to conduct electricity efficiently. It’s a bit like trying to drive on a bumpy dirt road when you really want a superhighway.
To make the electricity flow like a happy, gushing river, we sometimes add a tiny pinch of something extra to the water. This could be a little bit of salt (but not too much!) or a mild acid or base.
This little addition, called an electrolyte, doesn't get used up in the process. It just acts as a friendly facilitator, making it easier for the electrical current to zoom through the water and do its magic. It’s like giving our electrical current a super-speed boost!
The Power of Possibility: Why We Do This Atomic Dance!
So, why go through all this trouble to split water? Because hydrogen gas is incredibly special! It's touted as a fantastic, clean fuel for the future.
When hydrogen burns (or rather, "combines" with oxygen in a fuel cell), it releases a burst of energy. And guess what its only byproduct is? You guessed it: pure water vapor! It's like a perfectly circular, endlessly refreshing energy cycle.
Imagine cars running on this magical hydrogen, leaving nothing but a puff of pure water behind. Or powering homes and factories with this clean energy source. It’s a vision of a cleaner, brighter, and much happier planet!

The best part? If the electricity we use for this amazing water-splitting comes from renewable sources like solar panels soaking up sunbeams or wind turbines dancing in the breeze, we're making what's called green hydrogen!
"It's like turning water into a superhero fuel, powered by the sun and wind – a real high-five to Mother Earth!"
This means we're creating fuel with virtually zero carbon emissions, from start to finish. It’s a double win for the environment and a giant leap towards a sustainable future.
This isn't just science fiction; it's happening right now, in labs and pilot projects around the world. Scientists are constantly finding new and even more efficient ways to tickle those water molecules apart.
So, the next time you pour yourself a glass of water, take a moment to appreciate its hidden depths. It’s not just H2O; it’s a tiny, powerful chemical bond just waiting for its moment to shine, to be transformed into a fuel that could change our world.
It’s a truly magnificent marvel of microscopic engineering, right there in every single drop. Pure, clean hydrogen, ready to power our world with a friendly fizz and a whole lot of cheer!
