How Does Fire Extinguisher Work

Ever glanced at that bright red cylinder hanging on the wall in a public building, or maybe even in your own kitchen? Fire extinguishers are everywhere, silent sentinels watching over us. We see them, we know they're important, but have you ever really stopped to think about how they actually work?
They're kind of like silent superheroes, ready to spring into action at a moment's notice. But their power isn't magic; it's pure, elegant science! And it's actually a lot simpler and more fascinating than you might imagine.
The Fire Triangle: Understanding the Enemy
Before we dive into the gadget itself, let's talk about fire. Fire isn't just one thing; it's more like a party that requires three essential guests to keep going. Imagine it as a three-legged stool, and each leg is absolutely crucial:
Must Read
- Fuel: Something that can burn (wood, paper, gas, oil).
- Oxygen: The air around us that fire needs to breathe.
- Heat: Enough warmth to get the fuel hot enough to ignite and keep burning.
Take away just one leg, and poof – the stool (and the fire) comes tumbling down. This fundamental secret is what makes fire extinguishers so incredibly brilliant! They're designed to strategically attack one or more of these three elements.
Attack Plan #1: Cooling Things Down (Removing Heat)
So, how do our superhero cylinders tackle this fiery trio? One of the most intuitive ways is to simply cool things down.
Yep, we're talking about the classic water extinguisher. Water is fantastic at absorbing heat. When you spray water onto a fire, it rapidly turns into steam, soaking up a huge amount of thermal energy in the process.

Imagine pouring a bucket of cold water on a tiny campfire. It brings the temperature down so much that the fire just can't sustain itself anymore. It's like telling the fire, "Hey, your party's over, the vibe just isn't hot enough!" Simple, effective, and brilliant for things like wood, paper, and fabric fires (these are called Class A fires).
Attack Plan #2: Smothering the Flame (Removing Oxygen)
But what if water isn't the answer? What if you're dealing with an electrical fire, or something oily? You definitely wouldn't want to use water there, as it could spread the fire or conduct electricity!
That's where our extinguishers get a bit more cunning. They go after the oxygen. Fire needs air to breathe, just like we do. Cut off its air supply, and it suffocates. Think of it like putting a lid on a burning candle – the flame quickly vanishes as the oxygen runs out.
One common hero here is the Carbon Dioxide (CO2) extinguisher. When you pull the lever, a blast of incredibly cold CO2 gas rushes out. This gas is heavier than air, so it blankets the fire, pushing away the oxygen around it. No oxygen, no fire. It's a fantastic choice for electrical fires or flammable liquids (Class B and C fires) because it leaves no messy residue – great for sensitive equipment!
Another contender in the oxygen-deprivation game is foam. Foam extinguishers create a thick, stable barrier, a physical blanket that sits on top of flammable liquids, preventing oxygen from reaching the fuel and also helping to cool it.
Attack Plan #3: Chemical Warfare (Breaking the Chain Reaction)
Okay, so we've cooled fires and smothered them. Is there another trick up their sleeve?
You bet! Some extinguishers, especially the common ABC dry chemical extinguishers, employ a super-smart tactic. They don't just remove heat or oxygen; they actually interrupt the chemical reaction that makes fire possible.
Imagine fire as a series of tiny chemical dominoes falling over and over, sustaining the flame. These chemicals, usually a fine powder (often monoammonium phosphate or sodium bicarbonate), are like tiny ninjas that jump in and stop the dominoes from hitting each other.

The powder creates a barrier between the fuel and the oxygen, but more importantly, it interferes with the flame's ability to produce those reactive chemical intermediates needed to sustain itself. It's pretty high-tech stuff for a simple-looking canister, right?
That 'ABC' designation? It means it's versatile enough for Class A (ordinary combustibles), Class B (flammable liquids), and Class C (electrical) fires. Talk about an all-rounder!
The Inner Workings: A Quick Peek
So, how does all this magic powder and gas actually get out of the cylinder with such force?
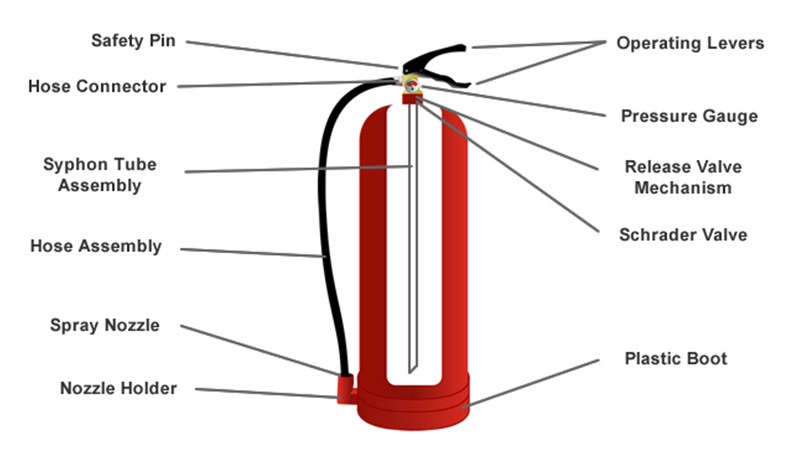
Most extinguishers use a stored pressure system. Essentially, there's a non-flammable gas (like nitrogen) under very high pressure inside the cylinder. When you press the handle, a valve opens, and this pressurized gas pushes the extinguishing agent – whether it's water, powder, or CO2 – out of the nozzle at impressive speed.

It's like shaking a soda bottle and then opening it, but way more controlled and, of course, way more useful!
The Awesome Bit
Isn't that wild? These humble red cylinders are packed with some serious science. They're not just random sprays; they're precisely engineered tools designed to exploit fire's fundamental weaknesses.
Next time you see one, give it a little nod of appreciation. It's a testament to human ingenuity, turning the destructive power of fire into something we can, quite literally, put out with a flick of the wrist. Knowing how they work makes them even cooler, right?
Stay curious, stay safe!
