How Does A Co2 Extinguisher Work

Ever wondered about those snazzy red cylinders chilling in the corner of offices and workshops? No, they're not giant cans of extreme energy drink (though that would be quite a morning pick-me-up!). We're talking about fire extinguishers, specifically the cool, calm, and collected CO2 variant. And today, we're going to peek behind the curtain and see how these unsung heroes actually work their magic. Spoiler alert: it involves a lot of science, but we'll keep it fun, I promise!
Meet the CO2 Extinguisher: Your Firefighting Friend
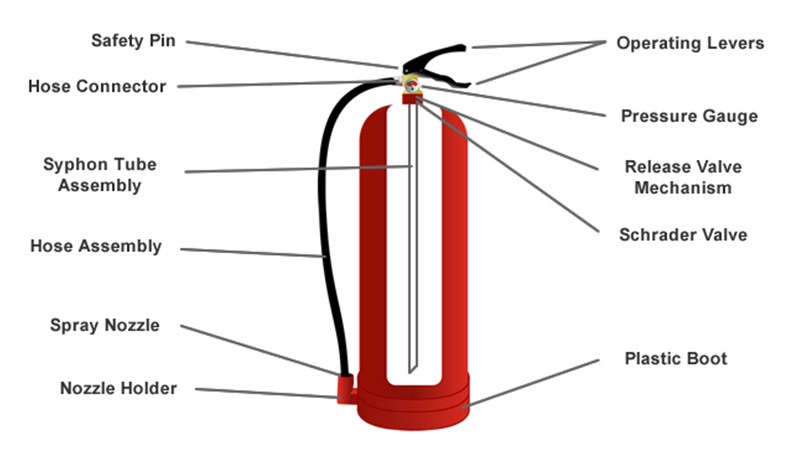
Imagine a superhero. Now imagine that superhero is red, has a horn-like nozzle, and carries its superpower in a super-pressurized tank. That's your CO2 extinguisher! It's especially good at tackling fires that involve electrical equipment or flammable liquids – basically, the kind of fires water would just make worse (and potentially give you a shocking surprise!).
The Secret Ingredient: Carbon Dioxide!
The star of our show is, you guessed it, carbon dioxide (CO2). Yes, the same stuff we breathe out and plants breathe in. But in a CO2 extinguisher, it's not just floating around casually. Oh no, it's packed in there under some serious pressure, turning it from a gas into a liquid. Think of it like a soda can before you open it – all that fizz is trapped CO2 waiting to escape!
Must Read
Under Pressure: The Liquid State
So, inside that sturdy red cylinder, the CO2 is compressed so much that it becomes a liquid. This is pretty clever because it means you can fit a lot more fire-fighting power into a smaller space. If it were just gas, the extinguisher would have to be enormous – like, "takes up half your kitchen" enormous. Not exactly practical for quick fire attacks, right?
The Big Squeeze: Releasing the Beast!
When you pull the pin (remember your P.A.S.S. acronym: Pull, Aim, Squeeze, Sweep!) and squeeze the handle, you're essentially opening a valve. This valve is like the floodgates for our superhero CO2. It's been waiting patiently, all cramped up as a liquid, and now it sees its chance to escape!

As the liquid CO2 rushes out of the nozzle, it experiences a dramatic drop in pressure. This sudden change is a bit much for our liquid friend, so it does what any self-respecting liquid under extreme pressure would do: it rapidly expands and turns back into a gas. But here's where it gets cool, literally.
The "Snow" Effect: A Chilly Blast!
This rapid expansion and transformation from liquid to gas causes the temperature of the CO2 to plummet – we're talking seriously cold, like minus 78 degrees Celsius (-108 Fahrenheit)! Some of the gaseous CO2 even freezes into tiny, icy particles, creating that distinctive white cloud or "snow" you see blasting out. It's not actually snow, of course, but rather a super-chilled, fine mist of dry ice. Don't try to catch it, unless you want an instant freezer burn souvenir!
How It Kills Fire: The Two-Pronged Attack
Now for the clever bit: how does this chilly gas actually put out a fire? It uses a dynamic duo of tactics:
1. Oxygen Deprivation (The Smothering Hug)
Fire needs three things to survive: heat, fuel, and oxygen (this is known as the fire triangle, for the science buffs among us). CO2 gas is much heavier than the air around it. So, when you blast it onto a fire, it descends and forms a blanket, effectively pushing away the oxygen that the fire needs to breathe. No oxygen? No fire! It's like gently but firmly tucking the fire into a permanent nap.
2. Cooling Effect (The Cold Shoulder)
Remember how we said that CO2 gas is ridiculously cold? Well, that extreme cold also helps by rapidly cooling down the burning material. By reducing the heat component of the fire triangle, it further hinders the combustion process. It's giving the fire the ultimate "cold shoulder" until it completely gives up.

When to Grab Your CO2 Buddy
CO2 extinguishers are superstars for Class B fires (flammable liquids like grease, oil, paint, gasoline) and Class C fires (electrical fires involving appliances, wiring, etc.). Because CO2 is a non-conductive gas and leaves no residue, it won't damage sensitive electronics or create a messy cleanup, unlike water or foam. Just remember, they're not the best choice for everyday materials like wood or paper (Class A fires) – for those, you'll want a different type of extinguisher.
A Word to the Wise (Still Fun, Though!)
While CO2 extinguishers are amazing, always remember to use them safely! Don't spray them at people (it's super cold and can cause frostbite!), and always aim at the base of the fire, sweeping the nozzle from side to side. And most importantly, always make sure you have a clear escape route. Safety first, even when battling fiery foes!
So There You Have It!
From a high-pressure liquid to a freezing gas, the CO2 extinguisher is a marvel of simple yet effective engineering, ready to zap those flames and save the day. It's a testament to how clever science can be, turning an everyday gas into a powerful fire-fighting tool. So next time you spot one, give it a respectful nod. It’s silently standing guard, keeping us all a little safer, and that’s something truly cool – much like the CO2 itself! Now go forth, be fire-aware, and maybe even impress your friends with your newfound extinguisher wisdom!

