H2so4 Naoh Balanced Equation

Ever wondered what happens when you mix some seriously potent chemicals? Like, the kind that wear lab coats and safety goggles potent? Well, let's dive into the fascinating world of chemistry and explore what happens when you combine sulfuric acid (H2SO4) and sodium hydroxide (NaOH). Don't worry, we'll keep it simple – no need for a degree in rocket science!
Think of it like this: sulfuric acid is like a grumpy old landlord, always demanding attention and easily irritated. Sodium hydroxide, on the other hand, is like a super chill mediator, always trying to balance things out. What happens when these two characters meet? That's where the magic of a balanced equation comes in!
What’s the Big Deal with a Balanced Equation?
So, what exactly is a balanced equation? Imagine baking a cake. You can’t just throw ingredients in randomly, right? You need the right amount of flour, sugar, eggs, and so on to get the perfect cake. A balanced equation is the same idea – it’s a recipe for a chemical reaction that ensures you have the same number of atoms of each element on both sides of the equation.
Must Read
Why is this important? Well, the Law of Conservation of Mass tells us that matter can't be created or destroyed in a chemical reaction. So, everything that goes in must come out, just in a different form! A balanced equation reflects this fundamental law.
The H2SO4 + NaOH Showdown
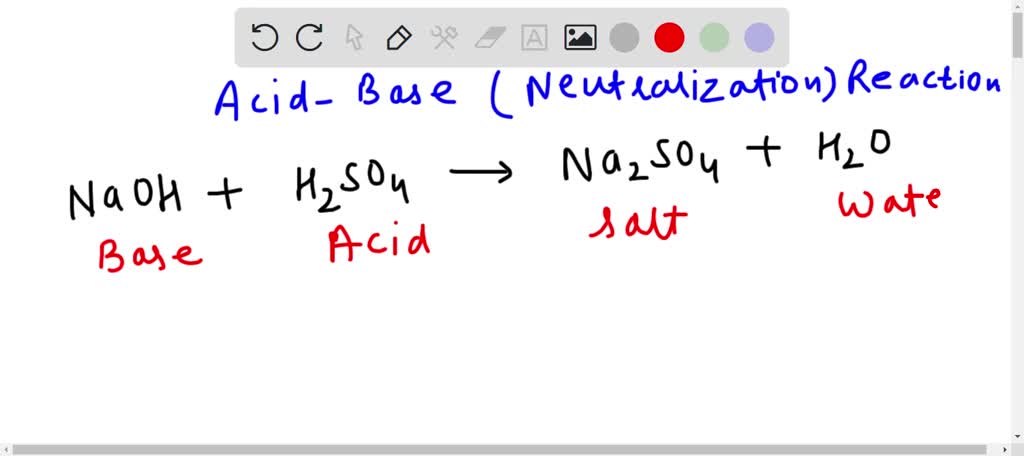
Okay, let’s get to the exciting part! When sulfuric acid (H2SO4) reacts with sodium hydroxide (NaOH), it's a classic acid-base neutralization reaction. Think of it like a superhero showdown where the "acid" villain is neutralized by the "base" hero. The result? Salt and water! (Don't go licking anything in a lab, though. Lab chemicals are a bit different from table salt!)
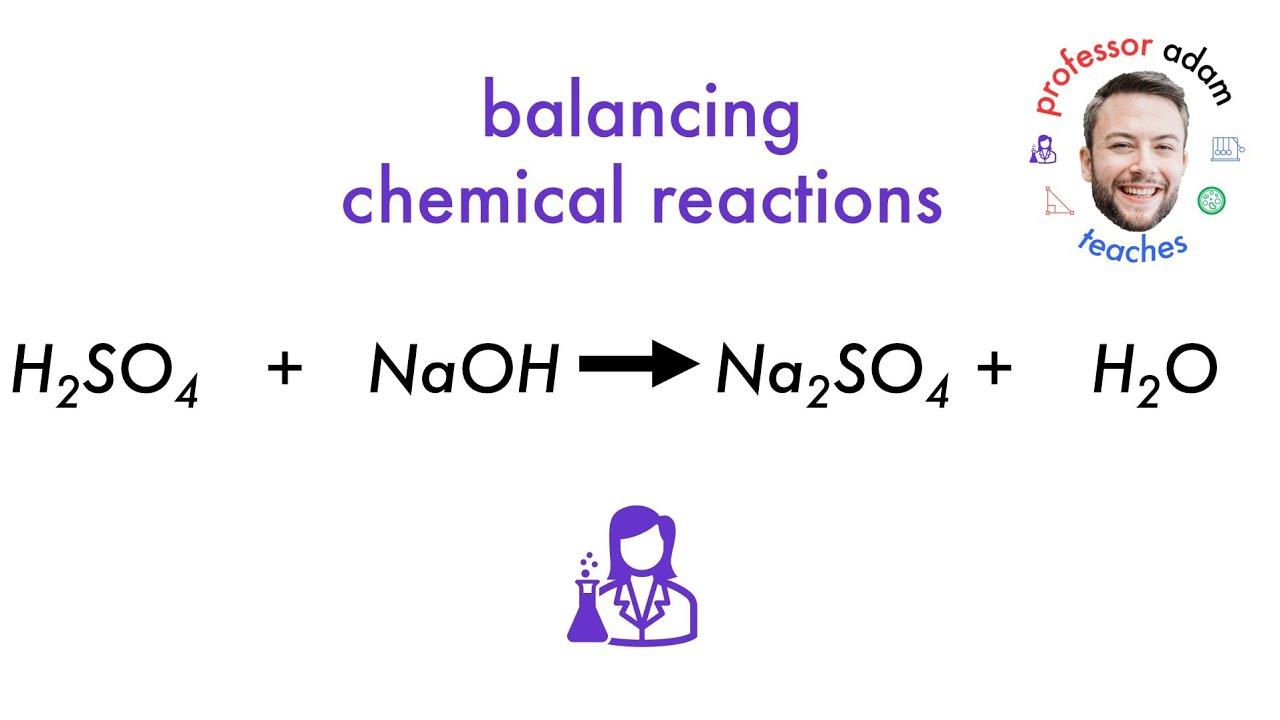
But what's the balanced equation for this superhero smackdown? It's not as simple as just throwing the formulas together. We need to make sure everything is accounted for.
Here's the balanced equation:
H2SO4 + 2NaOH → Na2SO4 + 2H2O

See those numbers in front of the NaOH and H2O? Those are called coefficients, and they're crucial for balancing the equation. They tell us the ratio in which the reactants (H2SO4 and NaOH) combine and the products (Na2SO4 and H2O) are formed.
Breaking it Down: Why the '2'?
So, why do we need a '2' in front of NaOH and H2O? Let's take a closer look. Sulfuric acid (H2SO4) has two hydrogen atoms (H). To neutralize both of those hydrogen atoms, we need two sodium hydroxide (NaOH) molecules. Each NaOH can "grab" one hydrogen atom to form water (H2O). That's why we end up with 2 water molecules (2H2O).
And what about the sodium (Na)? Since we used two NaOH molecules, we have two sodium atoms. These combine with the sulfate (SO4) from the sulfuric acid to form sodium sulfate (Na2SO4).

Consider this: * One H2SO4 molecule * Two NaOH molecules * Produce one Na2SO4 molecule * Produce two H2O molecules
Everything is neat and tidy, and the equation is balanced. We have the same number of hydrogen, sulfur, oxygen, and sodium atoms on both sides of the arrow!
Why Should You Care?
Okay, so balancing chemical equations isn't exactly the most thrilling activity. But understanding them is like unlocking a secret code to how the universe works! These reactions are happening all around us, from the digestion of food in your stomach to the production of electricity in a battery.
And more practically, if you're ever working in a lab (or even just trying to mix chemicals safely at home – always be careful!), knowing how to balance equations is essential for calculating the correct amounts of reactants to use and predicting the amount of product you'll get. Think of it as the ultimate recipe for successful chemical reactions.
Plus, it's pretty cool to impress your friends with your knowledge of chemistry, right? "Oh, that acid-base neutralization reaction? Yeah, I've totally got that balanced equation memorized." 😎
Final Thoughts
So, the next time you hear about sulfuric acid and sodium hydroxide, remember the balanced equation: H2SO4 + 2NaOH → Na2SO4 + 2H2O. It's more than just a bunch of letters and numbers – it's a reflection of the fundamental laws of nature and a key to understanding the fascinating world of chemistry. Keep exploring, keep questioning, and keep your curiosity alive!
