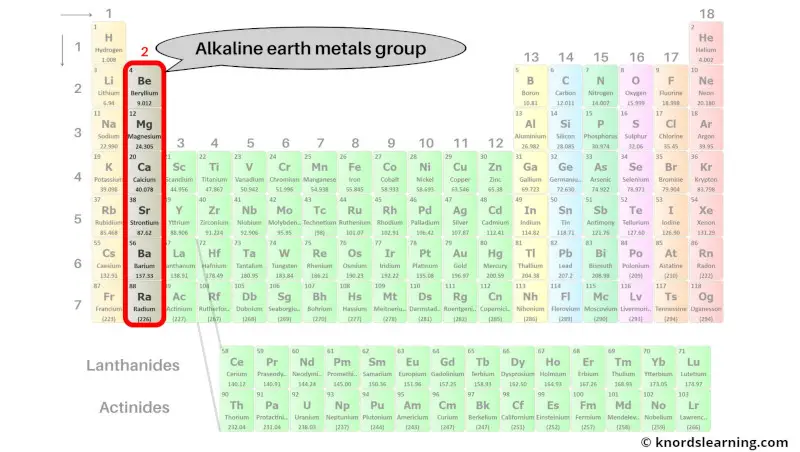
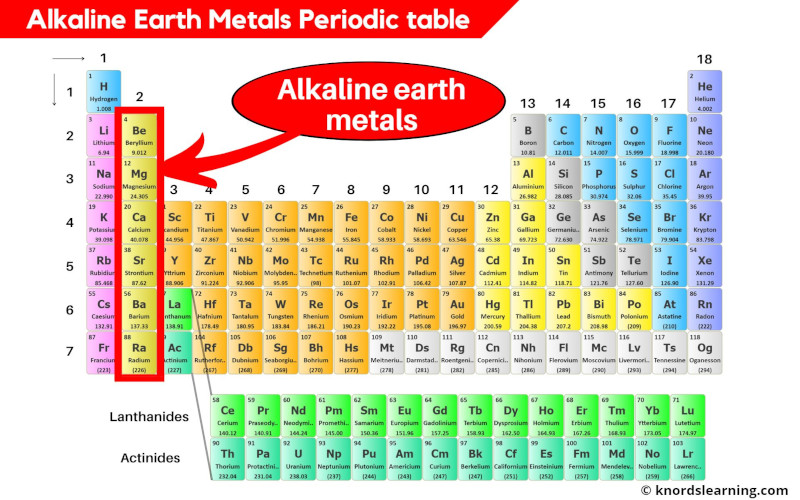
Group 2 Elements Alkaline Earth Metals
Ever heard of the unsung heroes of the periodic table? The ones that aren't flashy like their rowdy Group 1 cousins (we're looking at you, sodium, with your explosive personality!), but still bring a whole lotta awesome to the party? I'm talking about the Alkaline Earth Metals, the cool kids of Group 2!
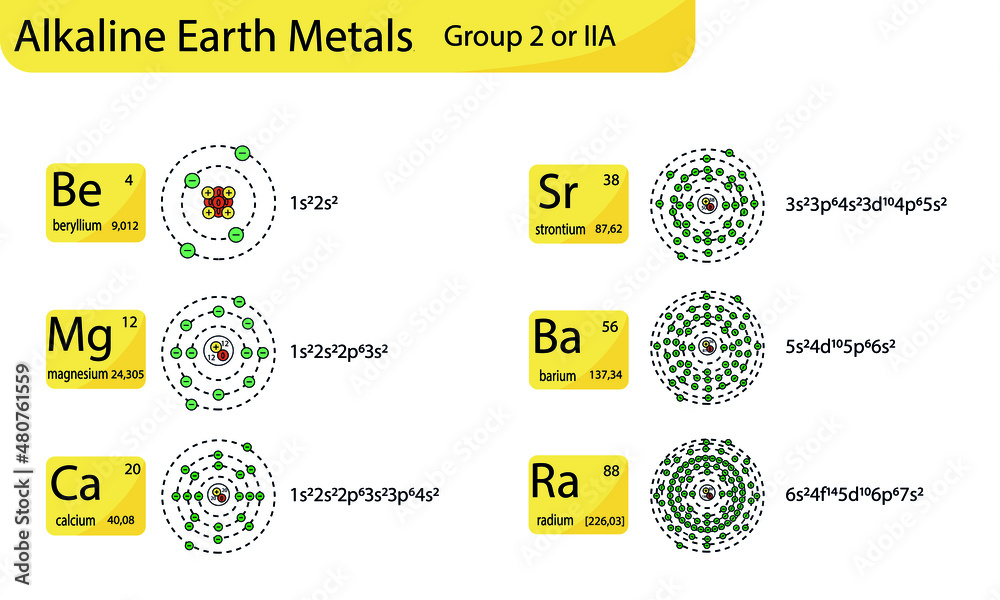
These guys – beryllium, magnesium, calcium, strontium, barium, and radium – they're not just hanging out on the periodic table looking pretty. Oh no! They're busy being the building blocks of, well, pretty much everything! Okay, maybe not everything, but a surprising amount!
Magnesium: The Multitasking Marvel
Let's start with magnesium (Mg). Think about that relaxing Epsom salt bath you take after a particularly brutal workout. Yep, that's magnesium sulfate doing its magic, soothing your aching muscles. But magnesium's talents don't stop there! It's also a key ingredient in chlorophyll, the stuff that makes plants green and allows them to perform photosynthesis, which basically means it's responsible for all the delicious oxygen we breathe! So, thank you, magnesium, for keeping us alive and relaxed. What a team player!
Must Read
Calcium: Bone-afide Superstar
Next up, the undisputed champion of strong bones and teeth: calcium (Ca). We all know that milk commercials hammered this one into our brains from a young age. But calcium isn't just about preventing osteoporosis (although, yeah, it's pretty darn good at that). It also plays a vital role in muscle function, nerve transmission, and blood clotting. It's basically the communication system and repair crew of your entire body! Without calcium, you'd be a wobbly, nervous, and possibly slightly leaky mess. No pressure, calcium!
Strontium: The Fireworks Fanatic
Now, let's talk about the element that likes to add a little sparkle to life: strontium (Sr). Ever watched a fireworks display and marveled at the brilliant red hues? That's strontium hard at work! Strontium salts are used to create those vibrant red colors in pyrotechnics. So, next time you're "ooh-ing" and "ahh-ing" at a fireworks show, remember to give a shout-out to strontium. It's the true color artist behind the explosions!

Also, and this is a bit of a quirky fact, strontium ranelate was once used as a medication to treat osteoporosis! It's like strontium was saying, "Hey calcium, I got your back! Let me help with those bones!". Sadly, it's use has been restricted due to potential side effects, but it was a noble effort!
Barium and Beryllium: The Underdogs (with Important Jobs!)
And then we have barium (Ba). You might not encounter it every day, but it's used in medical imaging! Specifically, barium sulfate is used in "barium swallow" tests, where patients drink a chalky liquid containing barium so doctors can get a clear view of their esophagus and stomach using X-rays. Okay, maybe not a super glamorous job, but definitely a vital one!
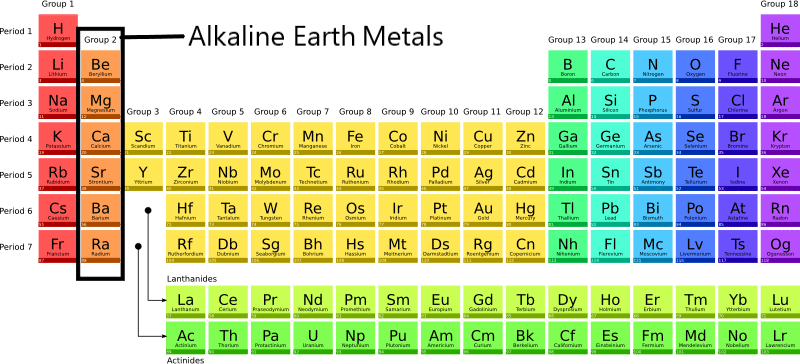
Finally, there's beryllium (Be). Beryllium is lightweight and strong, making it useful in aerospace applications. Think satellites and high-speed aircraft! It's also found in some specialized alloys that require high strength and thermal conductivity. So while it's less common in everyday life than, say, calcium, it's crucial in some pretty high-tech industries. It can be toxic in certain forms, so this is definitely one you want the professionals handling!
Radium: A Radioactive Reminder
And let's not forget radium! Now, this is where things get a little…intense. Radium (Ra) is radioactive, meaning it emits energy as it decays. In the early 20th century, it was used in glowing paints for watch dials (eek!) and even in some quack medical remedies. Nowadays, we know that prolonged exposure to radium is extremely harmful. It's a good reminder that even the most interesting elements need to be handled with respect and caution!
So, there you have it! A quick tour of the Alkaline Earth Metals. From building strong bones to lighting up the night sky, these elements are essential to our world. They might not always be the center of attention, but they're definitely worth appreciating! Now go forth and spread the word about the awesomeness of Group 2!