Formic Acid Lewis Structure
Okay, let's talk about something thrilling. Something that keeps chemists up at night (probably). I'm talking about... the Formic Acid Lewis Structure.
I know, I know. You're thinking, "Formic acid? Isn't that what ants spray?" Yes, genius, it is. And we're diving deep into its atomic handshake diagram today. Prepare yourself!
My Unpopular Opinion About It
Here's a confession: I kind of... like it. Gasp! I know, I know. People find it boring. But hear me out.
Must Read
It's simple. It's elegant. It's basically the "little black dress" of organic chemistry diagrams. So, you may hate me now!
Deconstructing the Delight (or Disappointment)
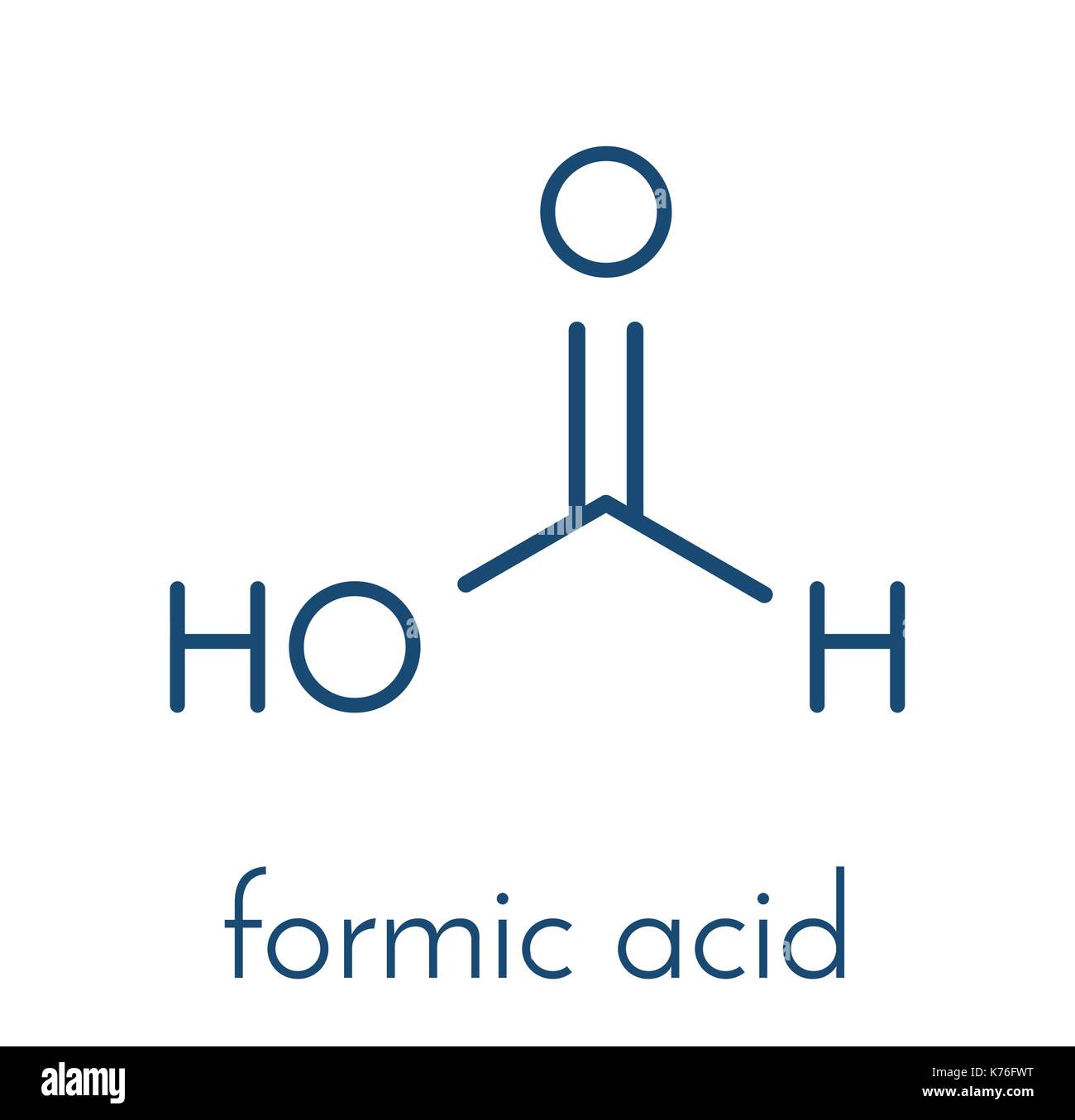
First, let's break down formic acid. Chemically, it's HCOOH. Or, if you're feeling fancy, methanoic acid.
Think of it like a tiny, angry robot with a mission. A mission to sting things. Just kidding (mostly).
Now, the Lewis structure. This is where the fun begins. We're drawing lines and dots like we're playing connect-the-atoms.
Carbon is at the center. Of course it is. Carbon always hogs the spotlight. Like that friend who always wants to be in the pictures.

It's double-bonded to one oxygen. That's where things get a little spicy. Double bonds mean extra electron sharing, extra commitment.
Then, it's single-bonded to another oxygen and a hydrogen. So, a balanced diet of single and double bonds. A happy carbon.
That second oxygen is also bonded to a hydrogen. A hydrogen hanging off the end. Super chill, just vibing.
The Lone Pairs: The Real MVPs
Now, the unsung heroes: the lone pairs. These are the extra electrons chilling on the oxygens.
They aren't bonded to anything. They're like the introverts at the party, observing everything from a safe distance.

Each oxygen has two pairs of these loners. Making sure the oxygens have their full octet. Gotta keep those atoms happy.
These lone pairs are super important! They contribute to the overall shape and reactivity of formic acid. More important than you thought!
Why I Find It Charming (Seriously!)
Here's where I defend my strange affection. The formic acid Lewis structure is so... fundamental.
It's a building block. It's a starting point. It's a reminder that even complex things are made of simple pieces.
Think of it like learning your ABCs. Essential, and a bit boring, but you can't write a novel without them. Formic acid is a chemistry ABC.

Also, it's symmetrical-ish. Which is pleasing to my eye. I appreciate the visual balance. Others prefer chaos, I prefer neatness.
And let's be honest, drawing it is kind of meditative. You meticulously place each dot and line. Instant mindfulness!
The Controversy (Because Everything is Controversial)
Okay, maybe "controversy" is too strong a word. But some people get the Lewis structure wrong.
They forget the lone pairs. Or they put too many bonds on the carbon. Don't be that person!
The carbon can only have four bonds! Remember the octet rule. It's not a suggestion, it's a law (of chemistry, at least).

A Call to Formic Acid Appreciation
So, next time you see an ant, remember formic acid.
Remember its humble Lewis structure. Remember the lone pairs, patiently waiting.
Maybe, just maybe, you'll find a little bit of charm in it too. Or, you know, just continue to think it's boring. I won't judge (much).
But I implore you, don't underestimate the power of a well-drawn Lewis structure! It could save the world! (Okay, maybe not. But it could help you pass your chemistry test.)
In conclusion, give formic acid Lewis Structure a second chance. You might be surprised.
It is not just a structure; it's an experience.
Thank you for coming to my TED Talk.
