Elements In Groups 3 Through 12 Are Called
Hey there, chemistry enthusiast (or just curious cat)! Ever wonder about those mysterious elements hanging out in the middle of the periodic table? You know, the ones that aren't quite as flashy as the alkali metals or as reactive as the halogens?
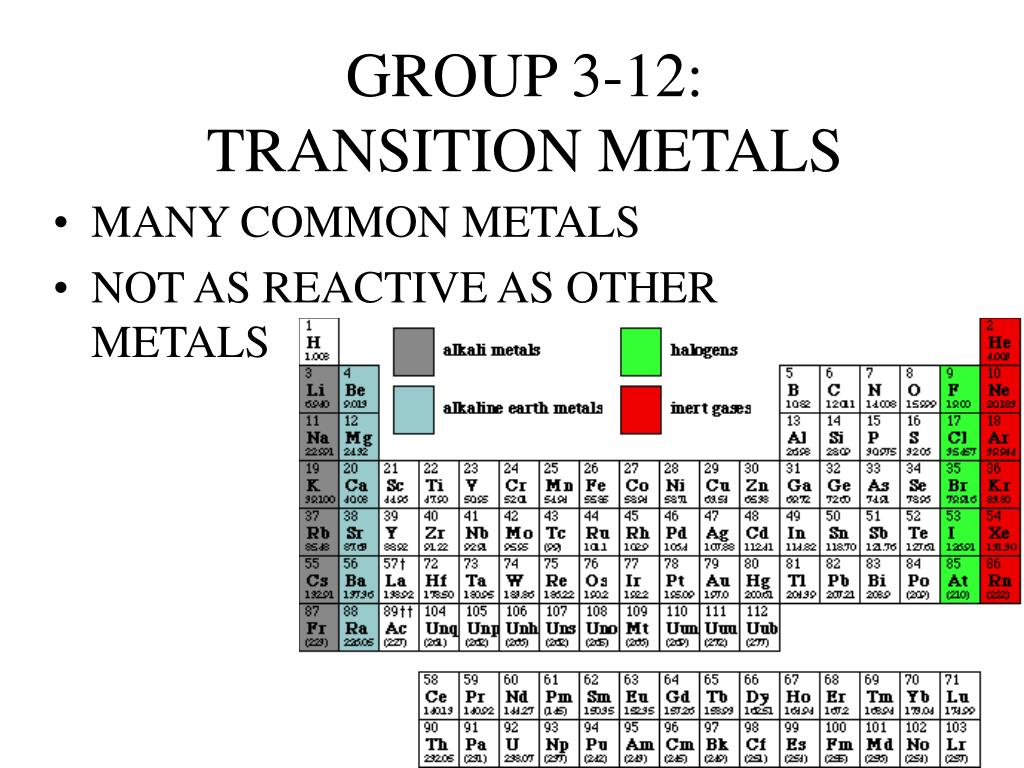
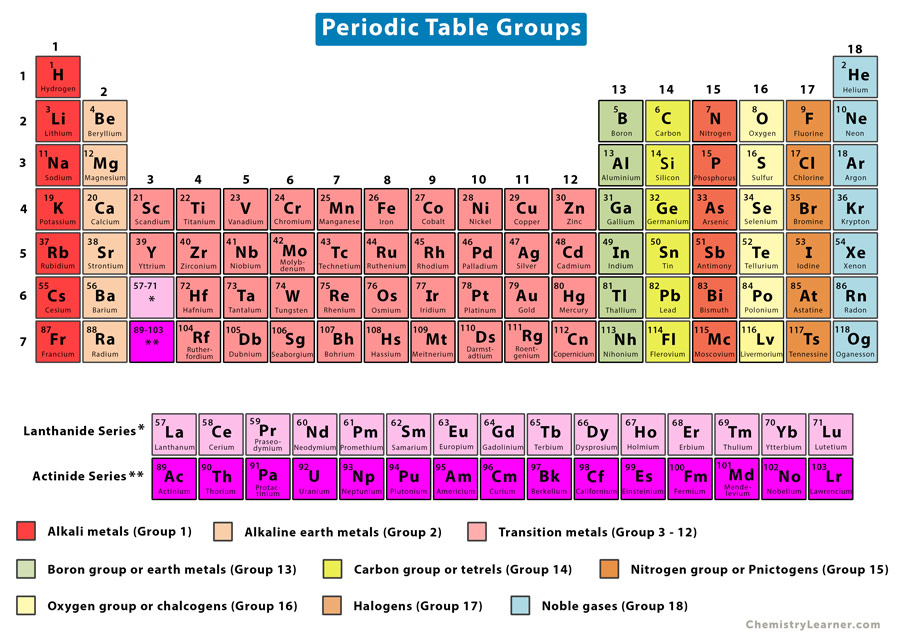
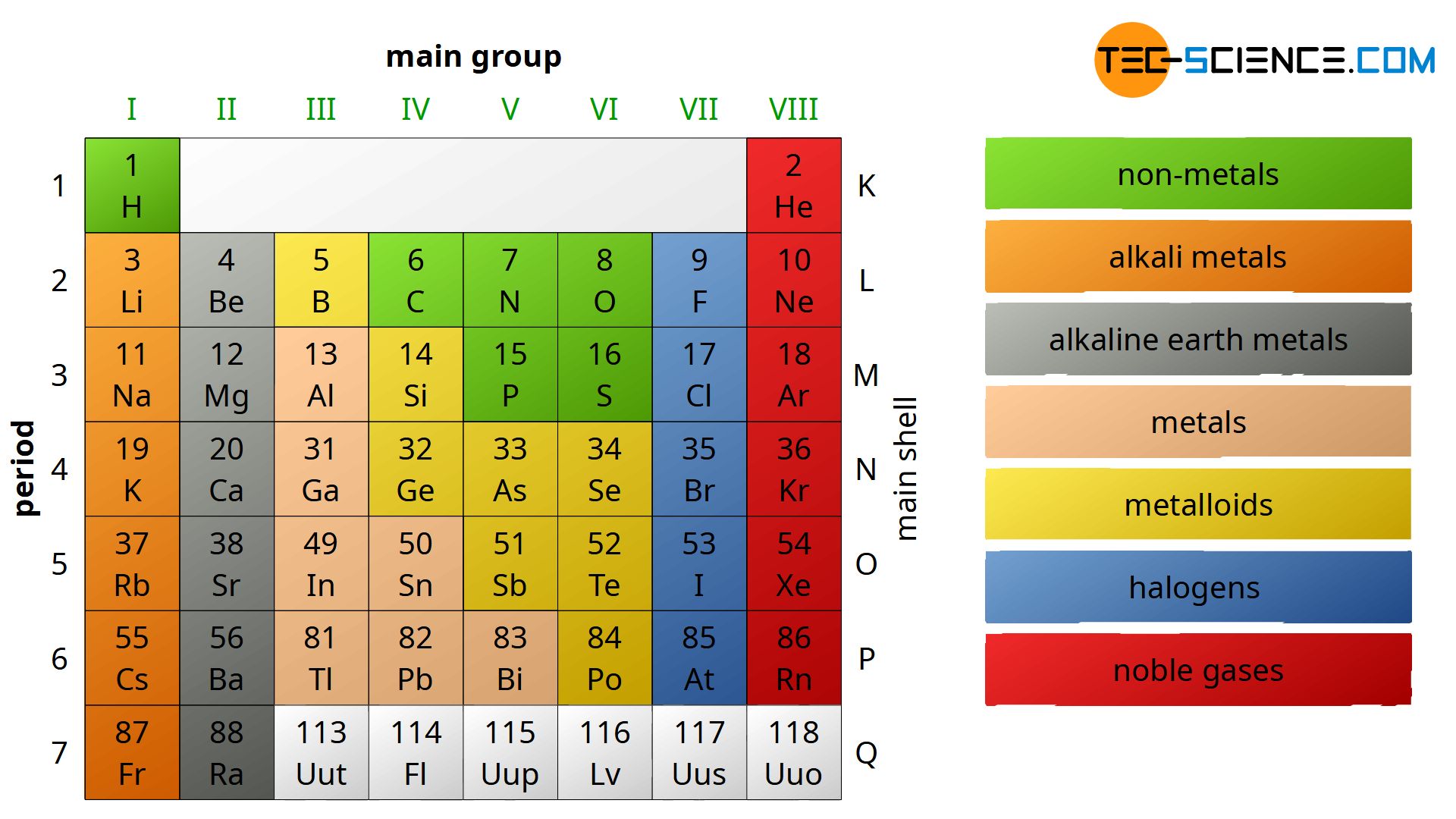
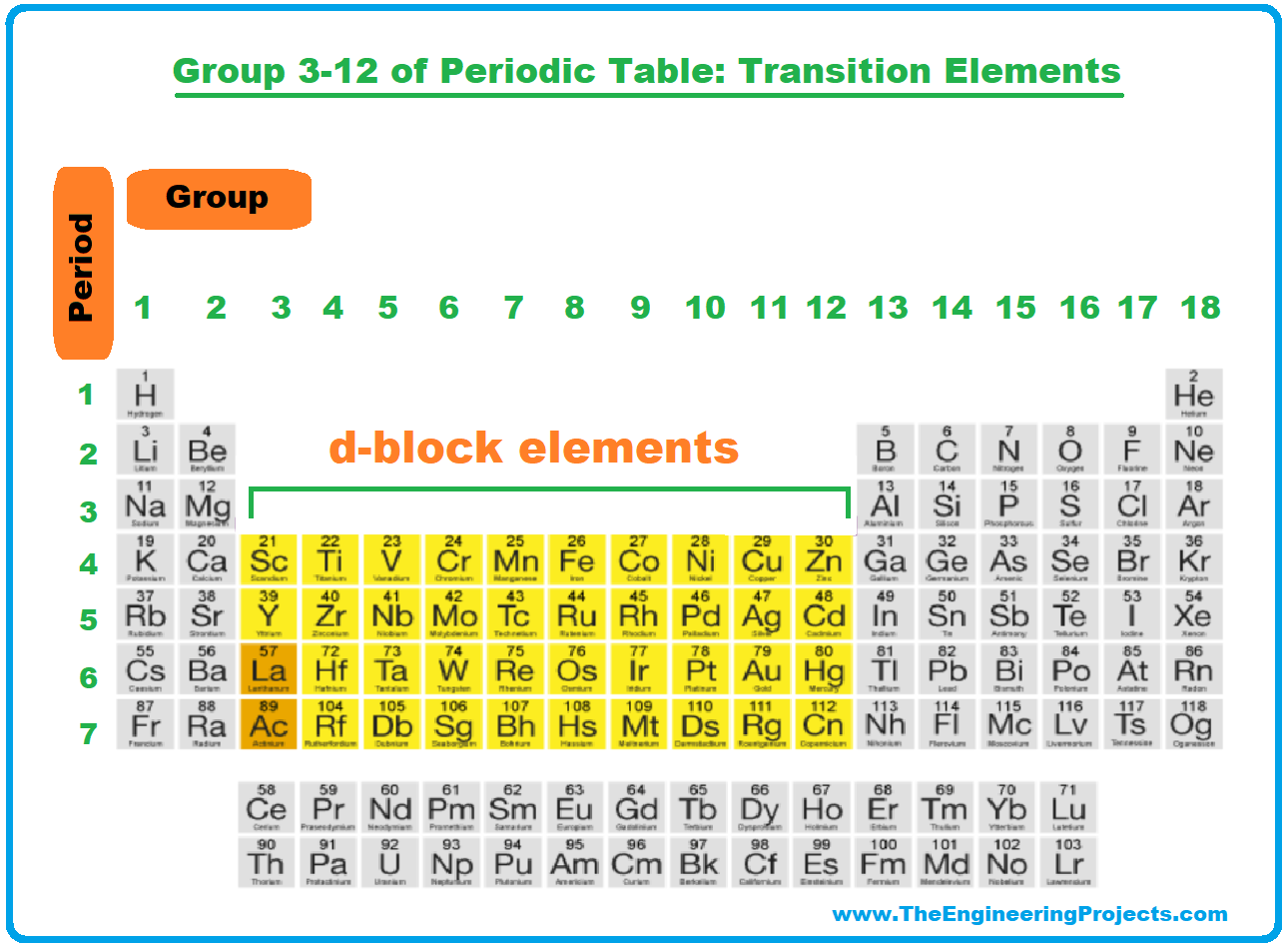
Well, buckle up! Today we're diving headfirst into the amazing world of… the transition metals! That's right, the elements in groups 3 through 12 are generally called transition metals.
Think of the periodic table as a bustling city. The alkali metals are the super-energetic folks living on the edge, always eager to make friends (react!). The halogens are the slightly dramatic, but equally engaging, neighbors on the other side. And the transition metals? They're the cool, collected folks hanging out downtown, forming the strong structural backbone of the city.
Must Read
What's So Special About Them?
Okay, so they're "cool and collected." But what does that actually mean? Why are they called transition metals anyway? Great question!
The name comes from their ability to "transition" between different oxidation states. Basically, they can happily lose a varying number of electrons when they bond with other elements. This flexibility gives them some pretty incredible properties.
For instance, iron can be Fe2+ (ferrous) or Fe3+ (ferric). Copper can be Cu+ (cuprous) or Cu2+ (cupric). It's like they have a whole wardrobe of electron outfits to choose from! This is due to the filling of the d-orbitals.
Think of it this way: imagine you’re going to a party. Some people can only wear one outfit – always formal, always casual. But the transition metals? They’ve got a closet full of options! They can rock a tuxedo, a t-shirt, or even a sparkly jumpsuit (if that’s their thing!).

Colorful Personalities
And speaking of sparkly, get ready for a visual feast! Many transition metal compounds are brilliantly colored. Think of the deep blue of copper sulfate crystals, the vibrant green of nickel compounds, or the rich purple of potassium permanganate.
These colors aren't just for show. They're a direct result of the way transition metal ions interact with light. The partially filled d-orbitals allow electrons to jump between energy levels when they absorb certain wavelengths of light. The light that isn't absorbed is what we see as color. Pretty neat, huh?
So, the next time you admire a beautiful gemstone, chances are it's the presence of a transition metal impurity that's responsible for its dazzling hue. Ruby gets its red color from chromium, while emerald owes its green to chromium and sometimes vanadium.
The Workhorses of the World
But the awesomeness doesn't stop at pretty colors. Transition metals are essential to so many aspects of our daily lives. They're the workhorses of the world!
Iron is the backbone of steel, the fundamental building material for everything from skyscrapers to cars. Titanium is incredibly strong and lightweight, making it perfect for airplanes and medical implants.
Gold and silver are prized for their beauty and conductivity, making them essential for jewelry and electronics. Platinum is a key component in catalytic converters, helping to reduce harmful emissions from vehicles.
Even within our own bodies, transition metals play vital roles. Iron is essential for carrying oxygen in our blood. Zinc is crucial for immune function and wound healing. Copper is involved in energy production and nerve function.
Quirky Facts to Impress Your Friends
Want to drop some knowledge bombs at your next trivia night? Here are a few quirky facts about transition metals:
Mercury is the only metal that is liquid at room temperature. Talk about being unique!
*Osmium is the densest naturally occurring element. A cubic foot of osmium would weigh over 1,400 pounds!
*Tungsten has the highest melting point of all metals. That's why it's used in light bulb filaments. So bright!
*Scandium was named after Scandinavia.
Why Should You Care?
So, why should you care about transition metals? Because they're everywhere! They're the building blocks of our world, the colors of our art, and the essential components of our bodies.
Understanding the properties of these elements can give you a deeper appreciation for the science behind everything around you. From the phone in your pocket to the blood in your veins, transition metals are playing a vital role.
Plus, it's just plain fascinating to learn about these versatile and colorful elements. So go forth and explore the wonderful world of transition metals! Your curiosity will be rewarded.
And remember, the periodic table isn't just a chart on a wall. It's a map to understanding the universe! Dive in, explore, and discover the amazing secrets that it holds. And don't forget, transition metals are in groups 3-12!
