Electron Withdrawing And Donating Groups
Hey there, science enthusiast! Ever wonder why some molecules are, well, a little clingy? Or super generous with their electrons?
Let's dive into the wacky world of electron-withdrawing groups (EWGs) and electron-donating groups (EDGs). Trust me, it's way more fun than it sounds!
What's the Deal with Electrons?
Okay, quick recap: Electrons are tiny, negatively charged particles. They're like the social butterflies of the atom world. They love to hang out with other atoms, forming bonds and making molecules.
Must Read
Now, some atoms are naturally greedy for electrons. They're like that friend who always eats the last slice of pizza. Others? Super chill. They're happy to share. This inherent difference is what drives the whole EWG/EDG thing.
Electron-Withdrawing Groups: The Energy Vampires
Think of EWGs as the energy vampires of the molecule world. They suck electrons towards themselves.
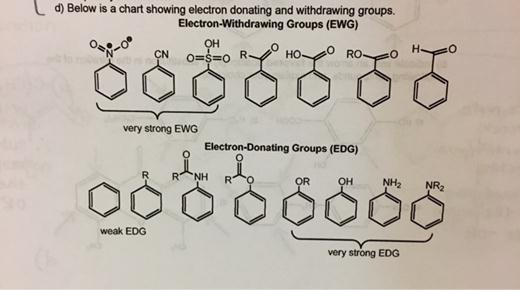
Examples? Halogens (fluorine, chlorine, bromine, iodine) are classic EWGs. They're like, "Gimme those electrons! I want them all!" Nitro groups (-NO2) are also notorious electron hogs.
Why do they do this? It's all about electronegativity, a fancy word for "how much an atom wants electrons." EWGs are highly electronegative.
Quirky fact: Fluorine is the most electronegative element. It's basically the electron equivalent of a black hole.

What happens when an EWG attaches itself to a molecule? It makes that part of the molecule electron-deficient. Imagine a tiny drought of electrons!
Electron-Donating Groups: The Generous Givers
On the flip side, we have EDGs. These are the philanthropists of the electron world. They donate electrons to the molecule.
Examples include alkyl groups (like methyl -CH3, ethyl -CH2CH3) and amino groups (-NH2). They’re like, “Here, have some electrons! You look like you need them!”
Why are they so generous? Well, they're not as electronegative as EWGs. They’re more willing to share the electron love.
When an EDG attaches itself to a molecule, it makes that part of the molecule electron-rich. A surplus of electrons! Party time!

Why Should You Care? (It's Actually Pretty Cool!)
So, why are we talking about this? Because EWGs and EDGs play a major role in how molecules behave.
They can affect:
*Reactivity: Whether a molecule is likely to react with something else.
*Acidity/Basicity: How easily a molecule donates or accepts protons (H+).
*Spectroscopic Properties: How a molecule interacts with light.

Think of it this way: EWGs and EDGs are like tiny switches that control a molecule's behavior. They can turn reactions on or off, make molecules more acidic or basic, and even change their color!
The Real World: It's Everywhere!
This stuff isn't just theoretical. EWGs and EDGs are everywhere in chemistry, from the drugs you take to the plastics in your water bottle.
For example, the effectiveness of certain pesticides depends on the presence of EWGs. They help the pesticide bind to its target more effectively.
And the color of some dyes is determined by the presence of EDGs. They change how the dye absorbs light, resulting in different colors.
Funny detail: Chemists often use these groups strategically to tune the properties of molecules. It's like playing molecular LEGOs!
It's All Relative: The Electron Tug-of-War
Keep in mind, whether a group acts as an EWG or EDG is relative. It depends on what it's attached to. Think of it as an electron tug-of-war. Who's pulling harder?
For example: Hydrogen (H) is often used as a reference point. Compared to hydrogen, most groups are either electron-withdrawing or electron-donating.
So, What's Next?
Hopefully, this has given you a fun and engaging intro to EWGs and EDGs. It's a fascinating area of chemistry with tons of practical applications.
The next time you see a chemical formula, try to spot the EWGs and EDGs. You'll be surprised at how often they show up. And who knows, maybe you'll even start thinking about how they affect the molecule's behavior. Because, let’s face it, understanding this stuff is secretly kinda awesome!
Remember, chemistry isn't just about memorizing facts. It's about understanding how the world works at a molecular level. And EWGs and EDGs are a key piece of that puzzle. Go forth and explore!
