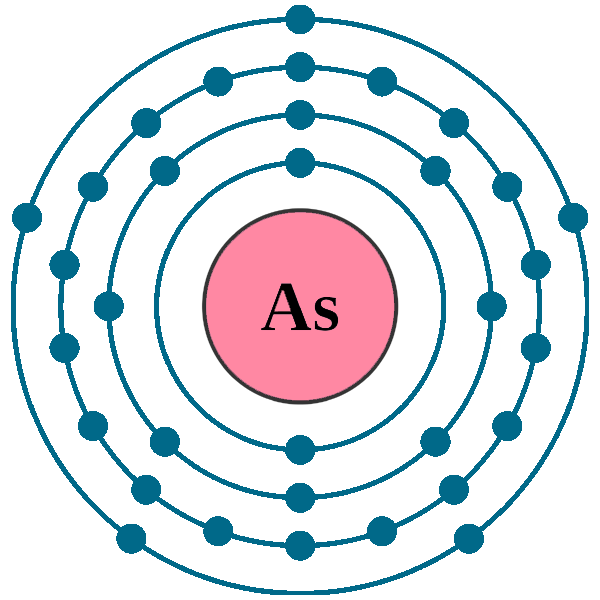
Electron Configuration For Arsenic

Arsenic. The name itself sounds like something a mischievous gnome might concoct in a bubbling cauldron. It's got a bit of a bad rep, historically used in all sorts of nefarious plots in old novels and movies. But let's forget the villainous associations for a moment and dive into something much less sinister (and honestly, way more interesting): Arsenic's electron configuration. Prepare to be… mildly amused!
Think of an atom like a miniature stadium, and electrons like eager fans trying to get the best seats. These seats aren't just scattered randomly; they're arranged in specific levels and sections. We call these levels "shells" and the sections "orbitals." Arsenic, being element number 33, has a whopping 33 electrons jostling for position. It's like trying to organize a flash mob, but with tiny, negatively charged particles.
The Electronic Seating Arrangement: Arsenic's Ticket to the Periodic Table
Now, here's where the electron configuration comes in. It's essentially a seating chart, telling us exactly where each electron is hanging out. Arsenic’s electron configuration is: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3. Okay, okay, that looks like alphabet soup, but let's break it down. Imagine it as a series of announcements over the stadium's PA system:
Must Read
"Attention fans! Two electrons to the 1s section!" (That's the innermost shell, closest to the nucleus – prime real estate!)
"Two electrons to the 2s section! Six electrons to the 2p section!" (The second shell is filling up nicely!)
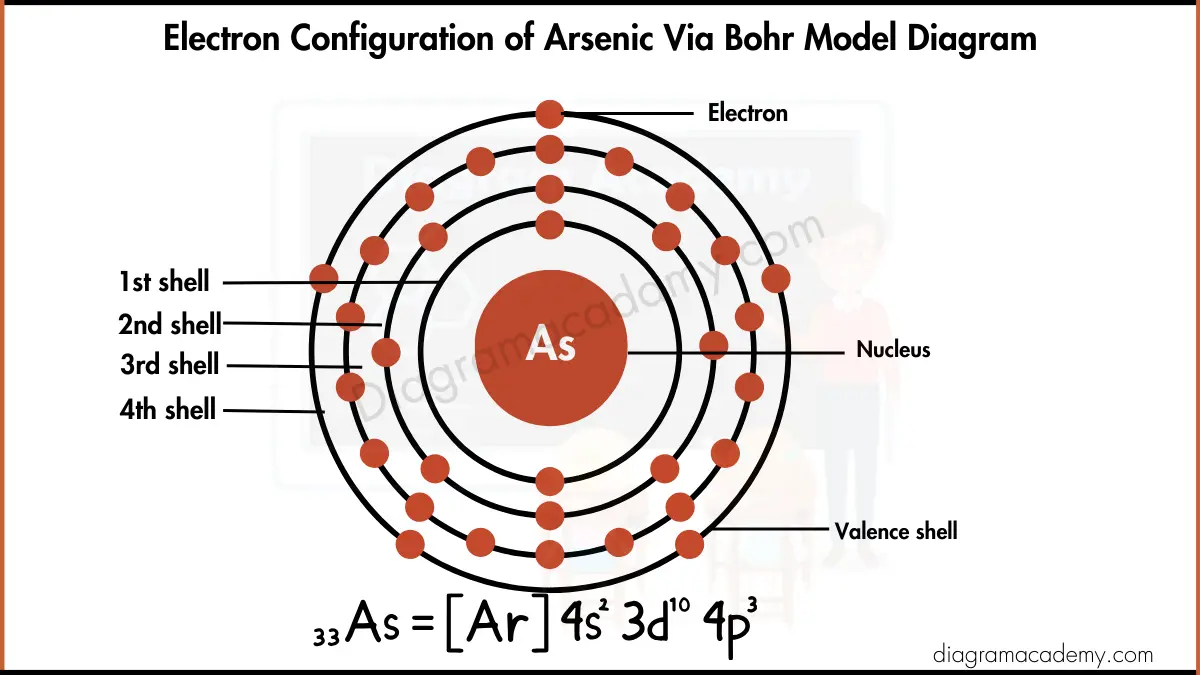
"Two electrons to the 3s section! Six electrons to the 3p section!" (We're getting further from the nucleus, but the energy is rising!)
"Two electrons to the 4s section! Ten electrons to the 3d section!" (Hold on, 3d fills after 4s?! It's like the stadium architect got confused with the blueprints! This is due to energy levels, but let's not get bogged down.)
"And finally, three electrons to the 4p section!" (These are Arsenic's valence electrons, the ones that really get involved in chemical reactions. They’re the social butterflies of the atomic world.)
See? Not so scary after all! It's just a carefully organized electron party. And the key takeaway? Those last three electrons in the 4p orbital are what makes Arsenic, well, Arsenic. They dictate how it bonds with other elements, and ultimately, influence its properties.

Why Should We Care About Arsenic's Electrons?
Besides being a fascinating insight into the structure of matter, understanding electron configuration allows us to predict how elements will behave. Those three valence electrons in Arsenic's 4p orbital? They’re the reason Arsenic often forms bonds with three other atoms, like a group hug involving four participants. This bonding behavior has profound consequences for everything from the formation of minerals to the action of certain poisons. Remember, knowledge is power (especially when it comes to potentially hazardous elements!).
Think about Arsenic in semiconductors, for instance. By tweaking the number of electrons in a material, we can control its ability to conduct electricity. This is the bedrock of modern electronics. So, while Arsenic might have a dark history, it also plays a vital role in the devices we use every day.

Moreover, the electron configuration helps us understand Arsenic's placement in the periodic table. It belongs to Group 15 (also known as the pnictogens – try saying that three times fast!), along with nitrogen, phosphorus, antimony, and bismuth. These elements share similar valence electron configurations, which explains why they exhibit some chemical similarities. It's like a family reunion, but with elements instead of eccentric relatives.
Arsenic: From Villain to… Misunderstood Hero?
So, the next time you hear about Arsenic, don't immediately think of murder mysteries. Instead, picture those 33 electrons, meticulously arranged in their atomic stadium, each playing its part in defining the element's properties. It's a reminder that even the most infamous elements have a complex and surprisingly beautiful story to tell, all written in the language of electron configuration. And who knows, maybe one day you'll be explaining the wonders of electron configuration to someone else, spreading the (slightly nerdy) joy! Maybe even use the analogy of the stadium with the fans, but don't forget to give credit!
And to end on a lighthearted note, if Arsenic could talk, maybe it would complain about always being typecast as the villain. "I'm more than just a poison!" it might lament. "I'm a crucial component in some very important technologies! Give me a break!" After all, even the most misunderstood elements deserve a little love (and a proper understanding of their electron configuration).
