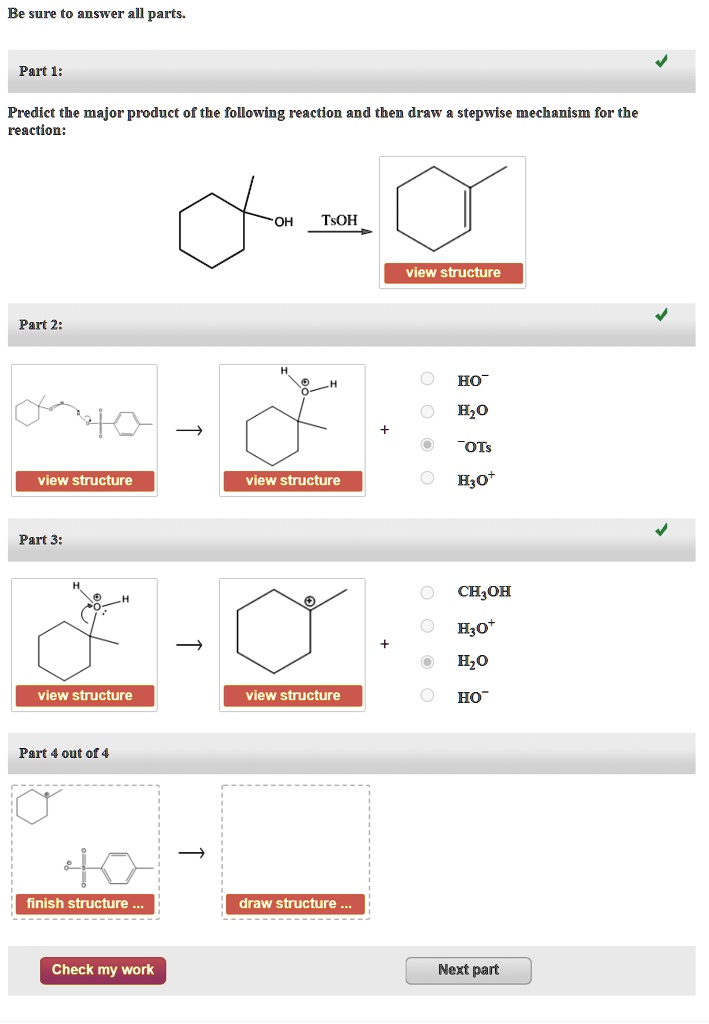
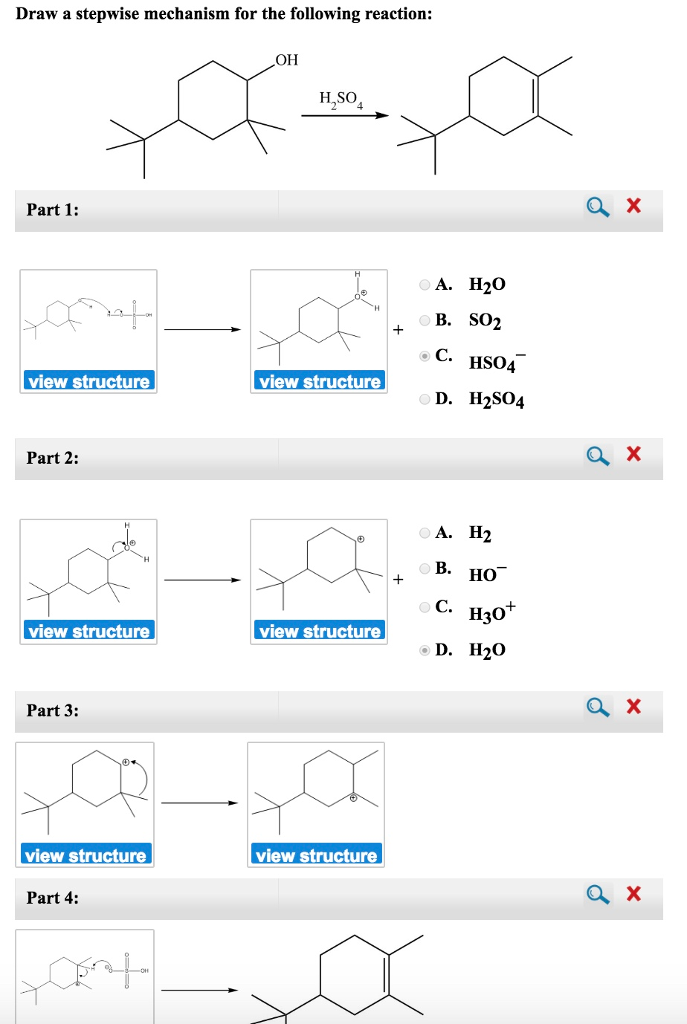
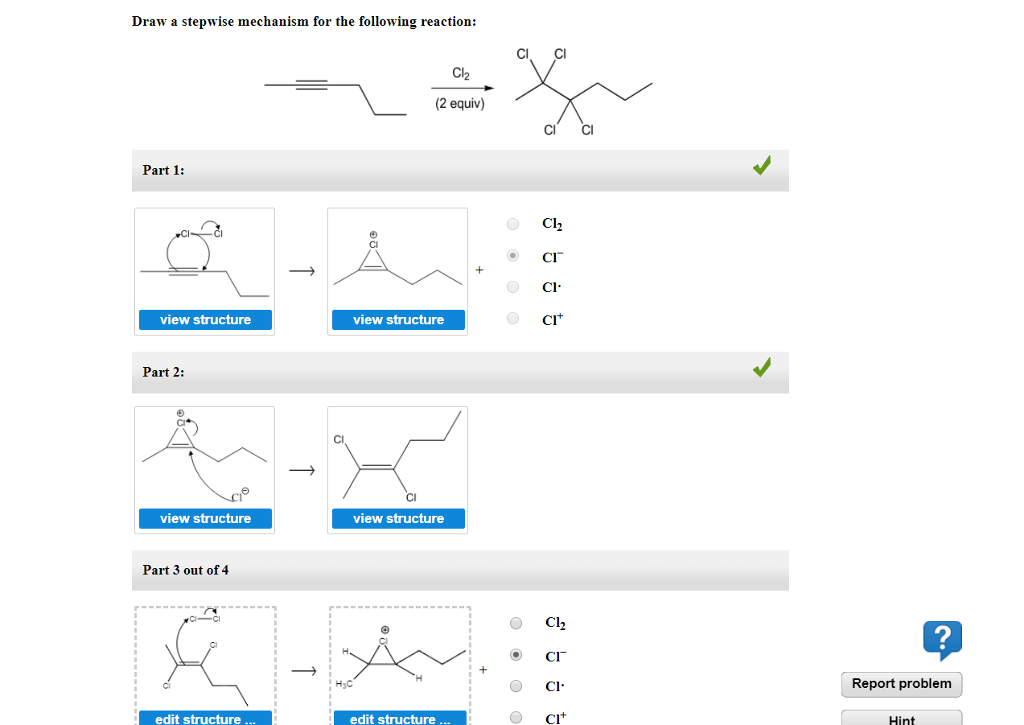
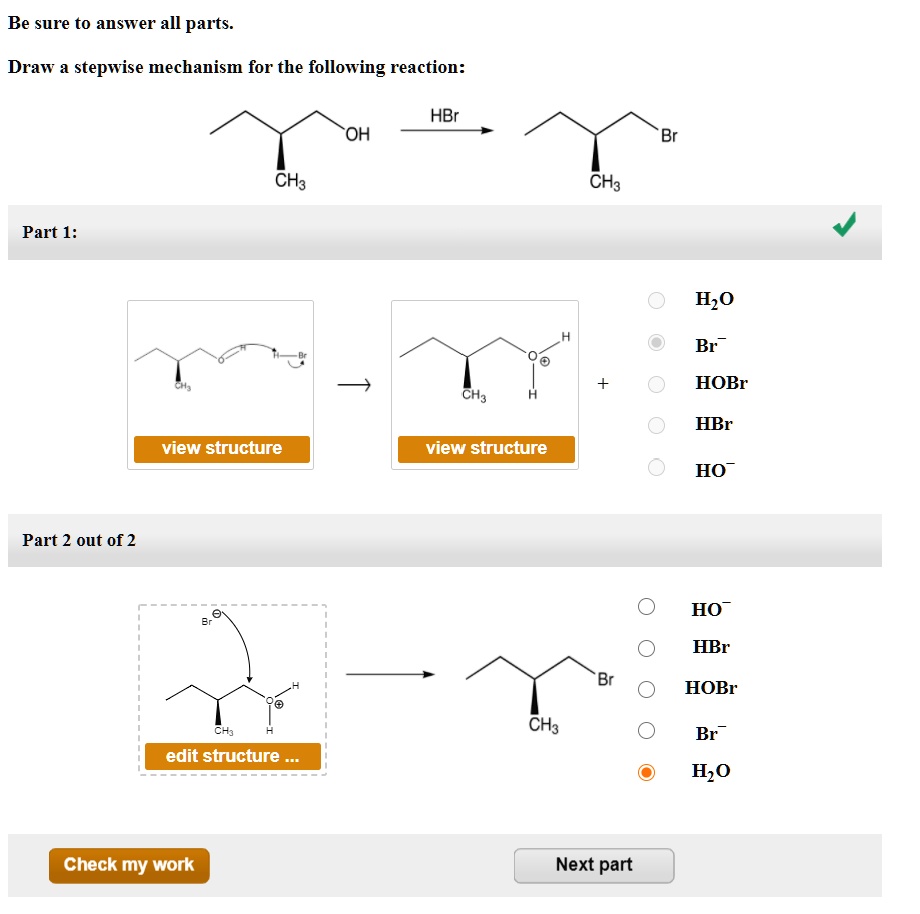
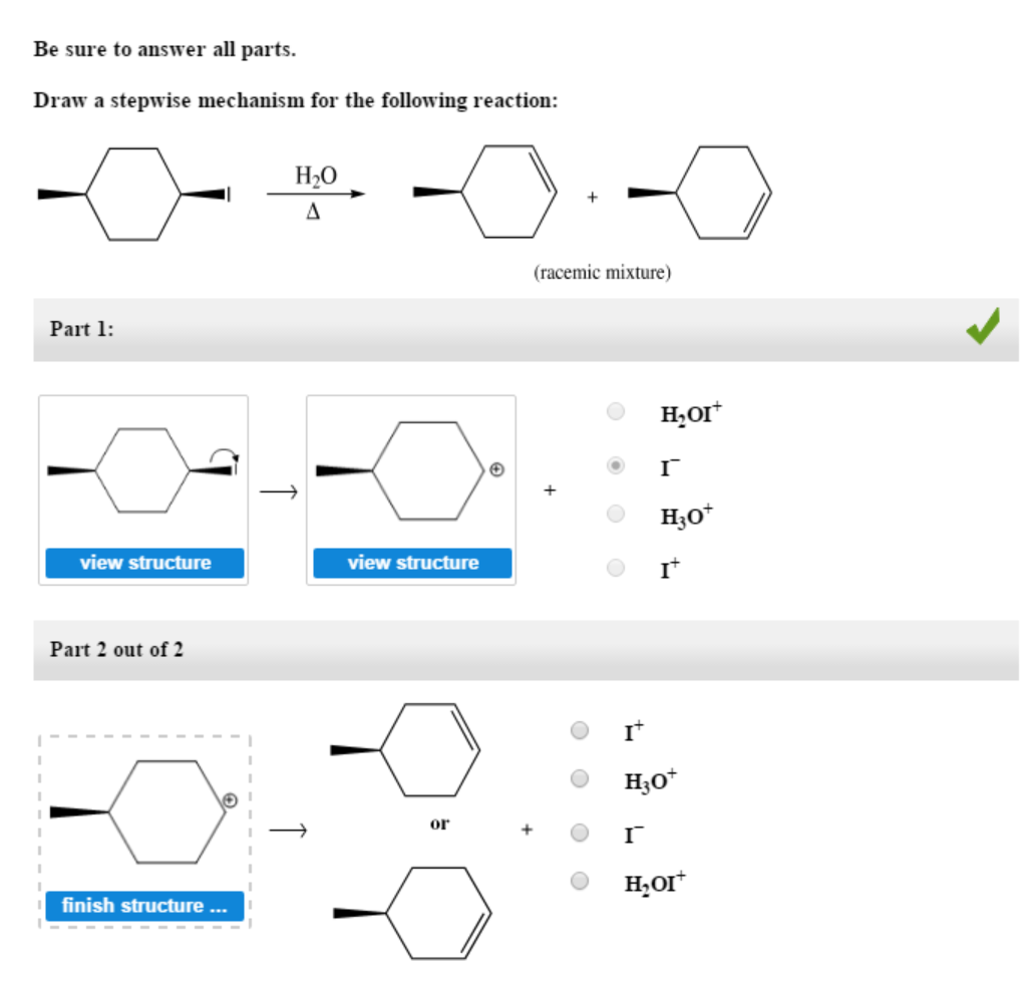
Draw The Mechanism For The Following Reaction
Hey there, chemistry comrade! So, you wanna tackle a reaction mechanism, huh? Awesome! It's like detective work for molecules, and who doesn't love a good mystery (especially one with electrons)? Let's dive in, but promise me you won't throw your textbook across the room, okay?
First things first: the big picture. What's reacting with what? Are we talking about an acid-base dance-off? Maybe a nucleophile wanting some action? Identifying your reactants and, more importantly, their personalities is crucial. It's like knowing your suspects in a whodunnit. You gotta know who's the aggressive one, who's likely to donate something, and who's just chilling in the corner.
Arrows are your friends! Seriously. In mechanism drawing, curved arrows show the movement of electron pairs. They are NOT suggestions. Think of them as tiny, electron-powered buses showing how electron density is shifting. ALWAYS start your arrows from a lone pair or a bond. Aim them at an atom or a bond where the electrons are heading. Accuracy counts, my friend. Sloppy arrows equal a chemistry frown-face.
Must Read
Now, let's break it down into steps. Most reactions don't happen in one magical poof. They're a series of elementary steps, each with its own electron shuffle. Ask yourself: What's the most electron-rich spot? Where's the positive charge (or partial positive charge) lurking? These are your starting points. Electrons are always attracted to positive (or partially positive) charges. It's like molecular magnetism! Who can resist?
Proton transfers are common. Don't be surprised if an acid or base pops into the picture. Watch out for those sneaky lone pairs! Remember, acids donate protons, bases accept protons. This dance of proton-y goodness often happens early in the mechanism, setting the stage for further shenanigans.
Breaking bonds: Think about bond polarity. Which atom is more electronegative? The more electronegative atom will hog the electrons, leading to a partial negative charge. When a bond breaks, those electrons typically go with the more electronegative atom. Boom! New lone pair. New negative charge (maybe!).
Forming bonds: Now we're cooking! A lone pair attacks an atom with a positive or partial positive charge. This can kick off a leaving group (more on those in a sec). Keep track of formal charges at all times. It's easy to lose track, but adding a charge where it doesn’t belong can change the entire meaning of the reaction. So, make sure you are adding your positive and negative charges in the appropriate spot. And don't forget to circle them!
Ah, leaving groups. These guys are like unwanted guests at a party. They get kicked out (with electrons, of course!) when something better comes along. Good leaving groups are stable anions (think halides – Cl-, Br-, I-). Think of them as the drama queens of the molecule world. They leave the party with flair and a good story to tell.
Don't forget about resonance. If a molecule has resonance structures, it can stabilize charges and influence the reaction pathway. Draw all possible resonance structures. Remember, resonance structures are not isomers – the atoms don't move, only the electrons do. It is just another way of drawing the same molecule.
Stereochemistry matters! Is your reaction going to create a chiral center? Does the molecule have any existing chiral centers? Pay attention to whether the reaction is stereospecific (one stereoisomer leads to a specific stereoisomer) or stereoselective (one stereoisomer is favored over another). Stereochemistry can make all the difference in the product you get, especially when we are talking about drugs.
Always double-check your work. Count atoms, count charges, and make sure everything makes sense. Does your mechanism lead to the observed product? If not, back to the drawing board, my friend. Consider, did you make a mistake when drawing your arrows? Did you forget to show the deprotonation of something?
And most importantly, practice, practice, practice! The more mechanisms you draw, the better you'll get. Eventually, you'll be able to predict reaction pathways with ease. It's like learning a new language – at first, it seems impossible, but with enough practice, you'll be fluent in the language of molecules.
So, grab a pen, paper, and maybe another cup of coffee. You got this! Now go forth and conquer those reaction mechanisms!