Does Hydrogen Have A Positive Or Negative Charge

Alright, folks, let's dive into a real head-scratcher: hydrogen's charge. Is it positive? Is it negative? Or is it playing some sort of neutral game?
The Obvious Answer (Or Is It?)
Textbooks scream "positive!" They shout about that single, lonely proton in the nucleus. It's positive, darn it! Case closed.
But hold on a second. Are we really going to let textbooks dictate reality?
Must Read
The Case for Positive (Yawn)
Hydrogen does have that proton. That's a fact. Protons are positive. Therefore, hydrogen, in its purest form, is positively inclined.
It's like saying a person with a winning lottery ticket is a winner. Sounds logical, right? But life, like hydrogen, is rarely that simple.
Why "Positive" is Too Simple
Here’s my unpopular opinion: hydrogen's charge is more complicated than a teenager's dating life.
It’s not just about that lone proton. It’s about context. It's about relationships. It's about the electron it usually hangs out with.
Enter the Electron: Hydrogen's Frenemy
Hydrogen usually chills with one electron. This electron, as we all know, is a tiny ball of negativity.
So, now we have a positive proton and a negative electron in the same tiny apartment (atom). What happens? Neutrality, baby!
But wait, there's more! Hydrogen is a social butterfly. It loves making friends (bonds) with other elements.
The Great Electron Tug-of-War
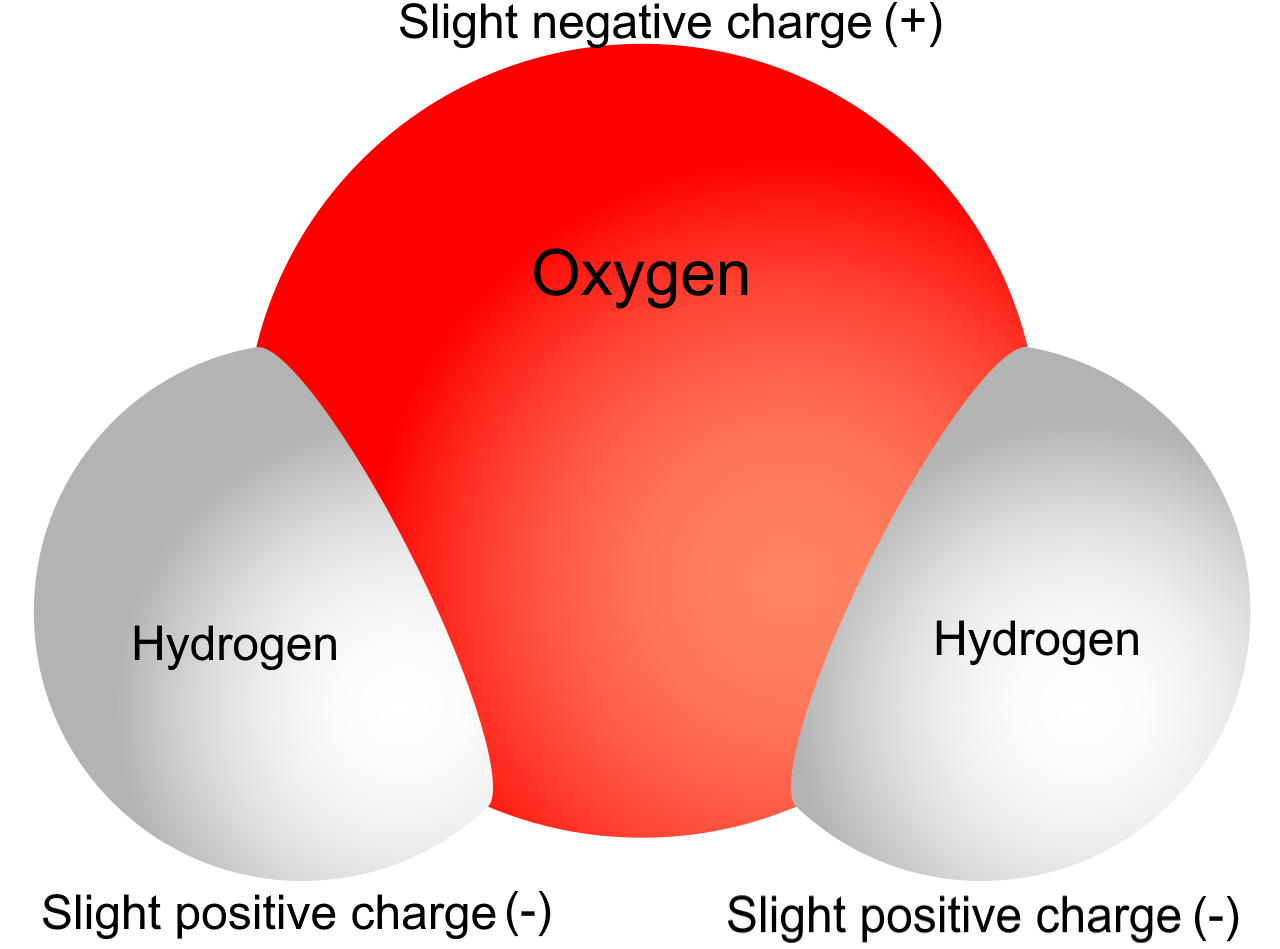
When hydrogen bonds with, say, oxygen in water (H2O), things get interesting. Oxygen is a bit of an electron hog.

It pulls the shared electrons closer to itself. This leaves hydrogen feeling a little…electron-deficient. A little…positive-ish.
This is called a partial positive charge (δ+). Notice the "partial." It's not a full-blown positive, just a hint.
Hydrogen's charge is like a mood ring. It changes depending on who it's hanging out with.
The Case for "Mostly Neutral"
Here's where I go rogue. I'm going to argue that hydrogen is, at its heart, mostly neutral.
Think about it. In its elemental form (H2), it's two hydrogen atoms sharing electrons equally. Balance. Harmony. Neutrality.
Even when it's bonded, it's often just a little bit positive or negative. A slight lean, not a complete flip.
My Unpopular Opinion: Hydrogen is Switzerland
Switzerland is famous for being neutral. Hydrogen, in my book, is the Switzerland of the periodic table.
It plays nice with everyone. It can be positive, negative, or neutral depending on the situation. It's all about diplomacy, baby!
What About Hydride (H-)? The Rebel!
Okay, okay, I hear you. What about hydride (H-)? That's hydrogen with an extra electron. Clearly negative, right?

Yes, hydride is definitively negative. It's the rebel of the hydrogen family. The one who dyes their hair black and listens to heavy metal.
But hydrides are relatively rare. They're the exception, not the rule. So, I'm sticking to my "mostly neutral" stance.
Hydride: The Exception That Proves the Rule?
Even hydride kind of proves my point. Hydrogen can be negative, but it takes effort. It needs to actively steal an electron to achieve that negativity.
It's not its natural state. It's a deliberate choice. Like choosing to wear socks with sandals.
So, What's the Verdict?
The "official" answer is that hydrogen has a positive nucleus due to the proton. Fine. I won't argue with the textbook definition.
But I will argue that it's a simplification. It's like saying a cat is just a furry mammal. True, but it misses the essence of the feline experience.
Hydrogen is more than just a positive charge. It's a chameleon. A shapeshifter. A master of disguise.
My Final (Probably Wrong) Answer
If you cornered me at a party and demanded a yes-or-no answer, I'd probably say hydrogen is mostly neutral.
It's a cop-out, I know. But it's the most honest answer I can give. It acknowledges the complexity of the element.
Plus, it's way more interesting than just saying "positive." Let's face it, "positive" is kind of boring.
Why This Matters (Sort Of)
Okay, so maybe this whole debate seems a bit academic. Does it really matter if hydrogen is positive, negative, or neutral-ish?
Well, understanding charge helps us understand how molecules interact. How chemical reactions happen. How life itself works.
Plus, it's a good reminder that things are rarely as simple as they seem. Especially in the world of science.
Embrace the Ambiguity!
So, the next time you think about hydrogen, remember that it's not just a positive proton. It's a complex, nuanced element with a complicated relationship with its electron.
Embrace the ambiguity! Revel in the uncertainty! And maybe, just maybe, agree with my unpopular opinion.
Or don't. I'm not the boss of you. But I still think I'm right.

Bonus Points: Hydrogen's Identity Crisis
Think about it: hydrogen is in Group 1 (alkali metals), but it's a nonmetal. It's also the most abundant element in the universe, but it's relatively rare on Earth.
It's like hydrogen is constantly asking itself, "Who am I, really?" A positive ion? A neutral atom? A negative hydride?
Maybe that's why it's so reactive. It's still trying to figure itself out. We can all relate to that, right?
Hydrogen: The Element We Can All Relate To
So, next time you're feeling confused or uncertain about your own identity, just remember hydrogen.
Even the simplest element in the universe has its existential crises. You're not alone!
And who knows, maybe embracing your own inner hydrogen – your own ability to be positive, negative, or neutral – is the key to a happier, more fulfilling life.
Now, if you'll excuse me, I'm going to go contemplate the meaning of life…and maybe try to steal an electron or two. For science, of course!
Thanks for reading!
