Does Bcl3 Have A Dipole Moment

Ever wondered why some things stick together, while others don't? Or why some molecules are drawn to water and others are repelled? A big part of the answer lies in something called a dipole moment. It's a fundamental property of molecules, influencing their behavior and interactions. And while it might sound like something confined to a lab, understanding dipole moments can actually unlock insights into the world around us. So, let's dive into whether Boron Trichloride, or BCl3, possesses this intriguing characteristic.
Simply put, a dipole moment arises when there's an uneven distribution of electrical charge within a molecule. Imagine a tug-of-war, where one side pulls a little harder. This creates a slightly negative end and a slightly positive end. The greater the difference in "pulling power" (electronegativity) between the atoms in a bond, and the more asymmetrical the molecule is, the larger the dipole moment. Molecules with significant dipole moments are known as polar molecules.
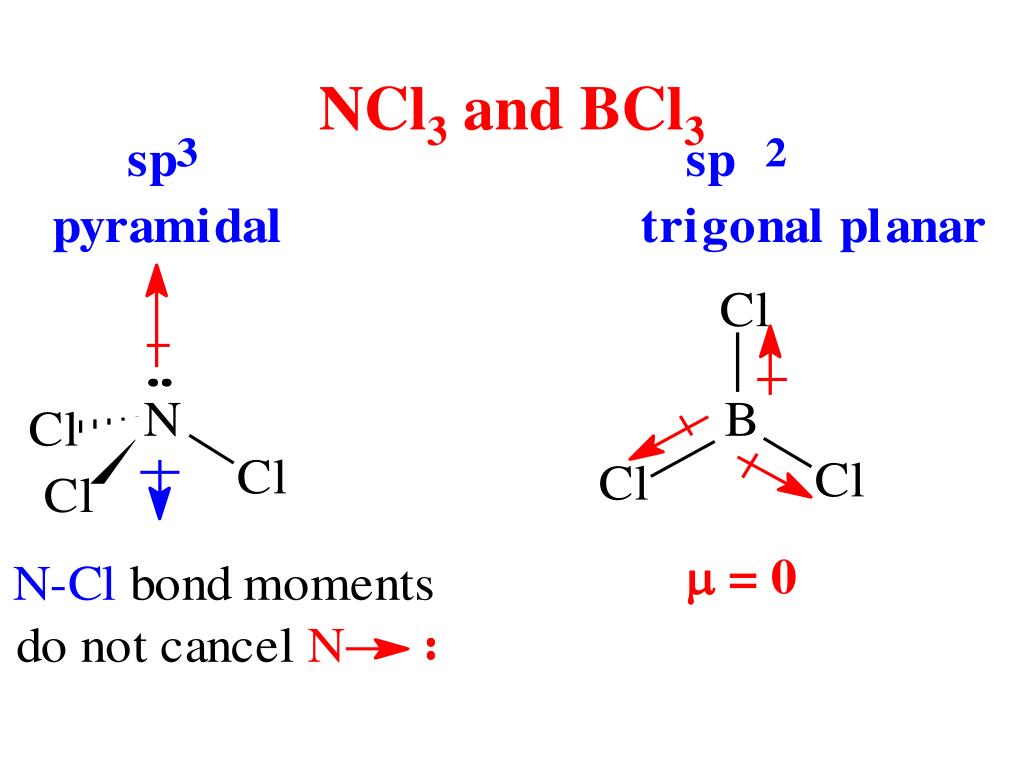
Now, for BCl3. Boron has three chlorine atoms bonded to it, forming a trigonal planar shape. This is where things get interesting. Chlorine is significantly more electronegative than Boron. This means each chlorine atom will pull the electrons in the B-Cl bond towards itself, creating a polar bond. However, the geometry of the molecule plays a crucial role. Because the three chlorine atoms are arranged symmetrically around the Boron atom, the individual bond dipoles cancel each other out. It’s like having three equally strong people pulling in different directions at equal angles – the net force is zero.
Must Read
Therefore, although each B-Cl bond is polar, the overall BCl3 molecule has no net dipole moment. It's a nonpolar molecule despite having polar bonds. This illustrates a key point: bond polarity doesn't automatically translate to molecular polarity. Molecular shape matters!
Why is this important? Well, dipole moments influence a molecule's physical properties. For example, polar molecules tend to have higher boiling points and melting points because the attraction between the positive end of one molecule and the negative end of another requires more energy to overcome. They are also typically more soluble in polar solvents like water. Understanding dipole moments can help predict how a substance will behave in different environments.

In education, understanding dipole moments is crucial in chemistry and materials science. Students learn about molecular geometry and electronegativity to predict molecular polarity. In daily life, the concepts apply, even if we don't realize it. For instance, detergents work because they have both polar and nonpolar regions, allowing them to interact with both water and grease. The effectiveness of certain medicines also depends on their ability to interact with specific molecules in the body, which is influenced by their dipole moments.
Want to explore this further? A simple way is to use online simulations that visualize molecular shapes and dipole moments. Many websites offer interactive tools where you can build molecules and see how the charge distribution changes. Another tip is to look up the electronegativity values of different elements. This will give you a sense of which bonds are likely to be polar. Finally, remember the importance of molecular geometry! Even seemingly simple molecules can exhibit complex behaviors depending on their shape and the distribution of electrons. Keep asking "why" and exploring the fascinating world of molecular interactions!