Difference Between Nuclear Fusion And Fission

Hey, grab a seat! Let’s talk about nuclear stuff. No, not THAT nuclear stuff… I mean the science-y kind. Specifically, nuclear fusion and fission. Sound intimidating? Don't worry, it's easier than parallel parking on a Tuesday afternoon.
Okay, so, what's the big difference? Think of it like this: one's about breaking things apart, the other's about smooshing them together. Got it? Good! Now for the slightly more complicated explanation (but still, pinky promise, not THAT complicated).
Nuclear Fission: The Breakup Artist
Fission, as the name sorta-kinda implies, is all about splitting atoms. We're talking heavy hitters like uranium, things that are already kind of unstable and just begging to fall apart. You basically bombard them with a neutron (think of it as a tiny atomic bowling ball), and BAM! They split!
Must Read
And when they split, they release a TON of energy. I mean, a LOT. Like, enough to power cities, or, you know… not so good things. (We're glossing over the not-so-good things for now). That energy then heats water, makes steam, spins a turbine, and voilá! Electricity! Who knew smashing atoms could be so...illuminating?
But wait, there’s more! That split also releases more neutrons. These neutrons then go on to split other atoms. Which release MORE neutrons. See where this is going? It's a chain reaction! (cue dramatic music). Control the chain reaction, and you have a power plant. Lose control? Well…let's just say things get messy. Like, REALLY messy.
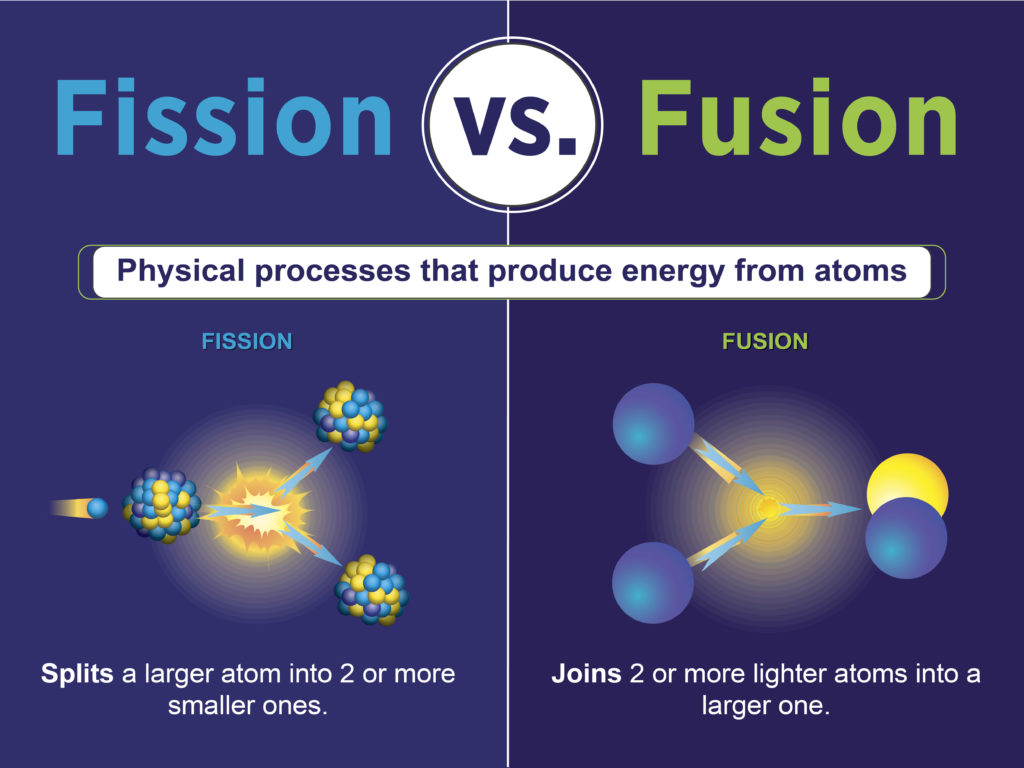
So, fission is basically taking something big and breaking it down into smaller, more manageable bits. Like breaking a giant chocolate bar into pieces...except with a whole lot more energy released. And, thankfully, fewer calories.
Nuclear Fusion: The Atomic Marriage
Now, let’s flip the script. Fusion isn't about breaking things; it's about combining them. Think of it as an atomic wedding, two tiny atoms becoming one! Specifically, we're usually talking about isotopes of hydrogen (deuterium and tritium, if you really want to impress your friends at your next atomic-themed party).
Here's the catch: Getting these atoms to fuse is like trying to force two magnets together with the same pole facing each other. They really, really don't want to do it. You need insane amounts of heat and pressure to overcome their natural repulsion. We're talking temperatures hotter than the sun! (Which, coincidentally, is where fusion happens naturally. Handy, huh?).

But if you can manage to get them to fuse, the energy released is even more mind-boggling than fission! And the best part? The byproducts are relatively harmless. Mostly helium, which is what makes balloons float. Think of it: clean, abundant energy and everyone gets a balloon! Win-win, right?
So, fusion is about taking two small things and fusing them together into something slightly bigger. Like... building a snowman? Okay, maybe not the best analogy. But you get the idea!
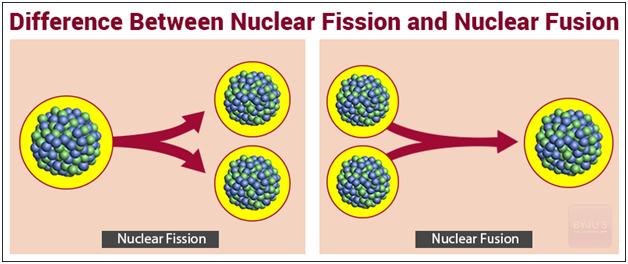
Fission vs. Fusion: A Quick Recap
Alright, let's recap, shall we? Pretend there's a quiz later (there isn’t, relax!).
- Fission: Splitting heavy atoms. Used in nuclear power plants (currently). Generates nuclear waste (boo!).
- Fusion: Combining light atoms. Happens in the sun. Clean energy (yay!), but really, really hard to achieve on Earth (currently).
So, while fission is our current nuclear workhorse, fusion is the holy grail. Scientists are working tirelessly (and probably fueled by copious amounts of coffee) to figure out how to make fusion a viable energy source. Imagine a world powered by the same process that fuels the sun! Pretty cool, huh?
Ultimately, both fission and fusion demonstrate the incredible power locked within the atom. One we've harnessed (with some drawbacks), the other we're still chasing. But hey, at least we have something to talk about over coffee, right?

