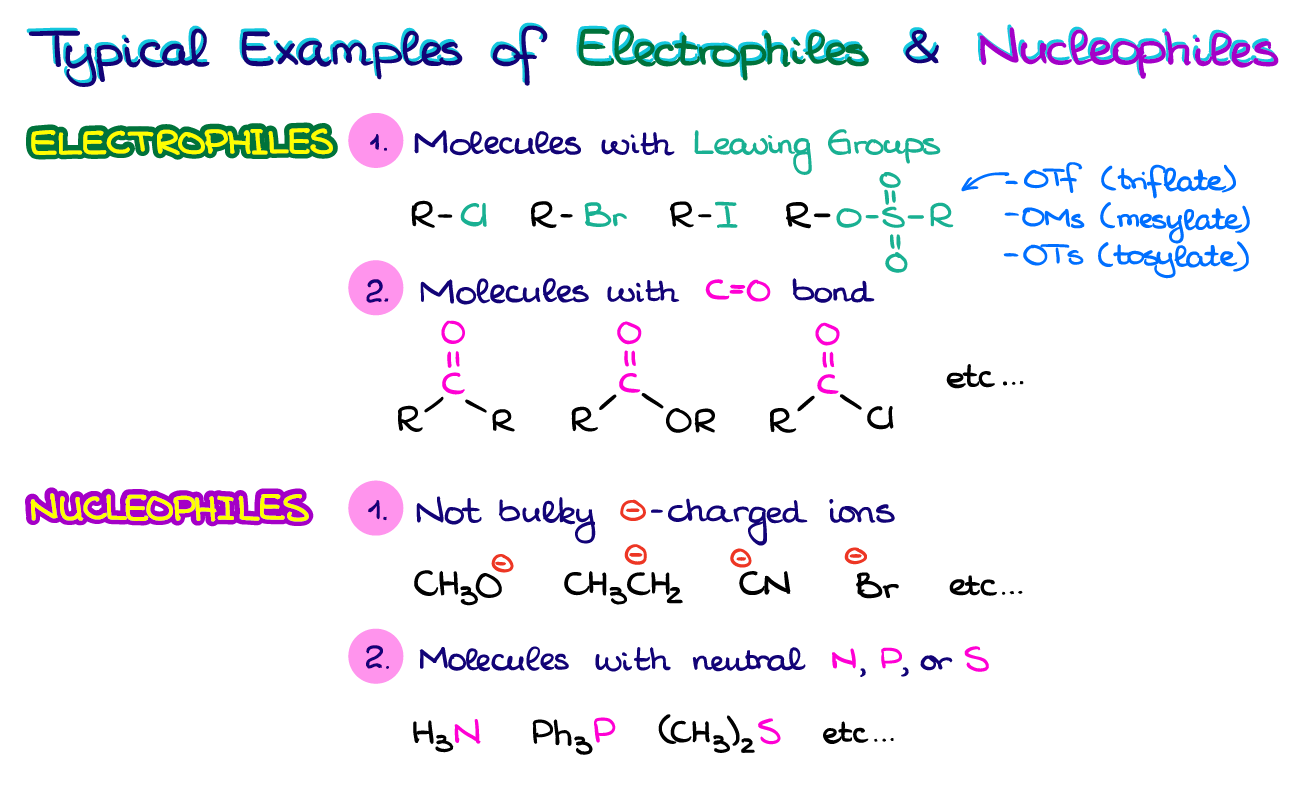
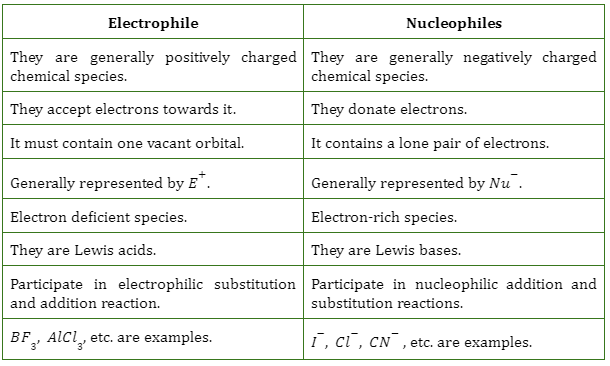
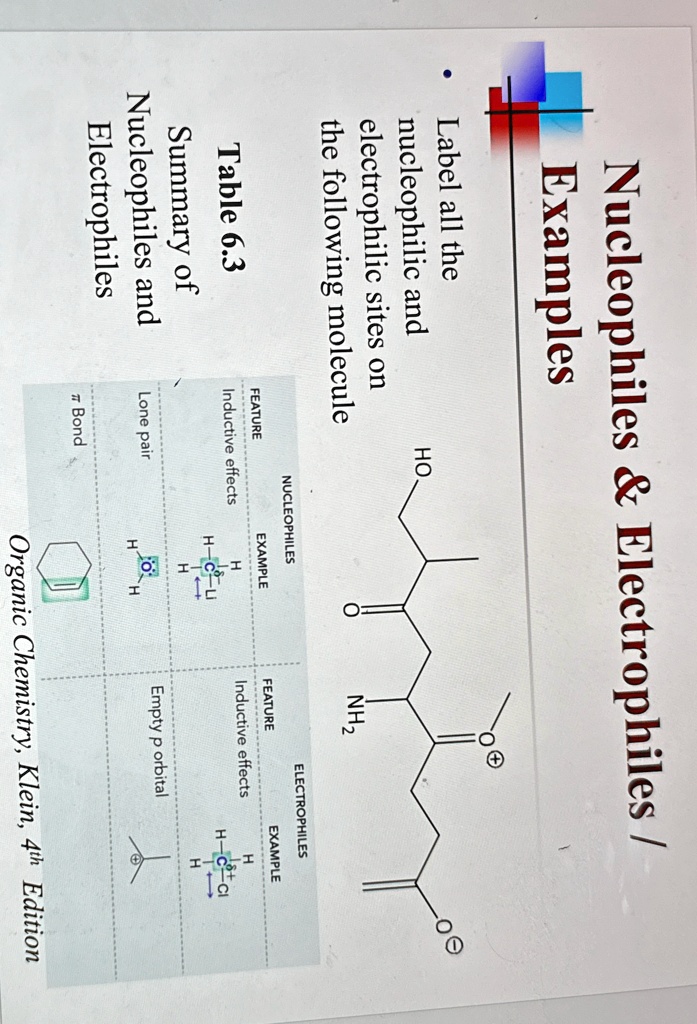
Define Nucleophile And Electrophile With Example
Imagine the world of chemistry as a giant dating pool. Instead of swiping right or left, molecules are either searching for love (electrons) or have plenty to give away.
This brings us to our star players: the nucleophiles and electrophiles. They’re the key to understanding countless chemical reactions, and surprisingly, they're kind of like characters in a sitcom.
The Nucleophile: The Generous Giver
Think of a nucleophile as the friend who always has an extra cookie, a spare charger, or a shoulder to cry on. They are generous with their electrons. Nucleophile literally means “nucleus-loving”.
Must Read
Since the nucleus of an atom is positively charged, these electron-rich friends are drawn to positive charges.
Basically, they're looking for someone (or something) positively charged to share their electron wealth with.
Examples of Nucleophiles
Water (H₂O) is a surprisingly good nucleophile. It has those lone pairs of electrons on the oxygen atom just itching to form a bond.
Another classic example is the hydroxide ion (OH⁻). It's essentially water with an extra electron, making it even more eager to donate.
Think of hydroxide ion as a super-generous water molecule with a serious case of "I have too many electrons!"
Ammonia (NH₃), the stuff that can sometimes make your cleaning supplies smell a bit funky, is also a nucleophile. The nitrogen atom has a lone pair, making it ready to mingle.
These nucleophiles are the unsung heroes of chemical reactions, constantly offering up their electrons to make new bonds.
The Electrophile: The Needy One
Now, let's meet the electrophile. These molecules are the opposite of nucleophiles.
They're like the friend who always needs to borrow money, a ride, or just a little bit of your time.
Electrophile literally means “electron-loving”. Because they’re electron deficient, they are always on the hunt for electrons.
Examples of Electrophiles
A simple example is a proton (H⁺), a hydrogen atom that has lost its electron. It's basically a bare positive charge desperately seeking some electron companionship.
Carbon dioxide (CO₂) can also act as an electrophile. The oxygen atoms pull electron density away from the carbon, making it slightly positive and vulnerable to nucleophilic attack.
Imagine CO₂ as a molecule that's trying to be stable but secretly yearns for more electron attention.
Another electrophile is a carbocation, a carbon atom with only three bonds and a positive charge. Carbocations are notoriously unstable and react quickly with any nearby nucleophile.
They’re like the desperate singles of the molecular world, ready to latch onto the first electron pair they can find.
The Chemical Romance: Nucleophile Meets Electrophile
The magic happens when a nucleophile and an electrophile meet. It’s a classic story of opposites attracting.
The nucleophile, rich in electrons, donates some of its electron density to the electrophile, which is electron-deficient.
This sharing of electrons creates a new chemical bond, forming a new molecule.
Think of it as a molecular marriage, where the nucleophile gives a precious gift (electrons) to the electrophile, creating a lasting bond.
For example, the hydroxide ion (OH⁻) can attack a carbon atom in methyl bromide (CH₃Br). The hydroxide ion donates its electrons to the carbon, kicking off the bromine atom and forming methanol (CH₃OH).
This is a simple example of a substitution reaction, where one group (the bromine) is replaced by another (the hydroxide).
These reactions are fundamental to organic chemistry and are used to synthesize a vast array of molecules, from pharmaceuticals to plastics.

Why Should You Care?
You might be thinking, "Okay, that's interesting, but why should I care about nucleophiles and electrophiles?"
Well, these reactions are happening all around you, all the time. They are the basis of life itself.
From the digestion of food in your stomach to the synthesis of proteins in your cells, nucleophiles and electrophiles are constantly interacting.
Understanding these fundamental concepts can give you a deeper appreciation for the complexity and beauty of the natural world.
Plus, it's kind of fun to think of molecules as having personalities and desires, searching for their perfect electron match.
The Surprising Humor
Sometimes, the reactions between nucleophiles and electrophiles can be surprisingly amusing. Imagine a bulky nucleophile trying to squeeze its way into a crowded molecule to attack an electrophilic center. It's like trying to parallel park a bus in downtown Manhattan.
Or picture a carbocation, desperately flailing around, trying to grab any electrons it can find before it gets neutralized. It’s like a toddler reaching for a cookie just out of reach.
These reactions aren't always smooth and elegant. Sometimes, they're messy, chaotic, and even a little bit awkward.

The Heartwarming Aspect
But beyond the humor, there's something almost heartwarming about the interaction between nucleophiles and electrophiles.
It's a story of giving and receiving, of need and fulfillment. It's a reminder that even at the molecular level, there's a fundamental drive to connect and form bonds.
The nucleophile, with its generous spirit, reaches out to the electrophile, offering it the electrons it so desperately needs. In return, the electrophile provides stability and a new purpose for the nucleophile.
It’s a beautiful example of symbiosis, where two different entities come together to create something greater than the sum of their parts.
In Conclusion
So, the next time you hear the words nucleophile and electrophile, don't be intimidated.
Remember the generous giver and the needy one, the molecular dating pool, and the constant search for electron companionship.
Chemistry isn't just about complicated equations and obscure lab experiments. It's about the fundamental interactions that shape our world, interactions that are surprisingly relatable, humorous, and even a little bit heartwarming.
And who knows, maybe understanding nucleophiles and electrophiles will even help you find your own perfect match, whether it's a molecule or a friend!
