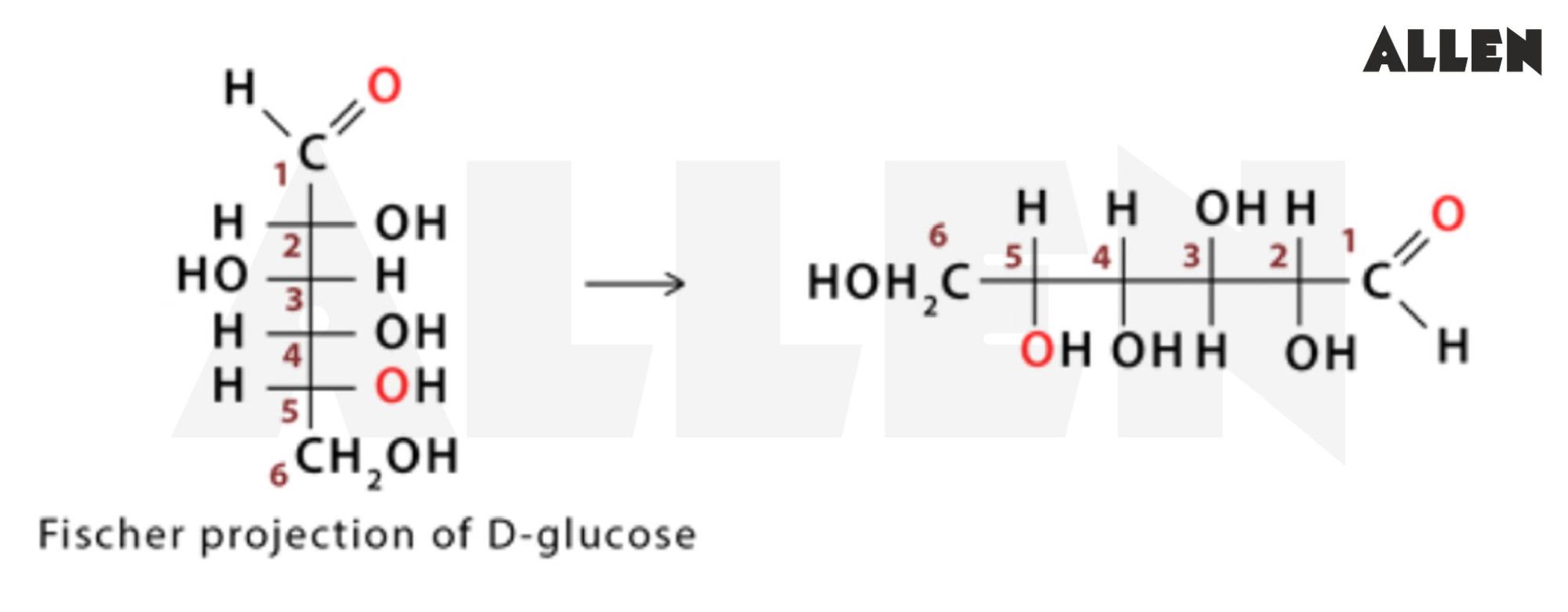
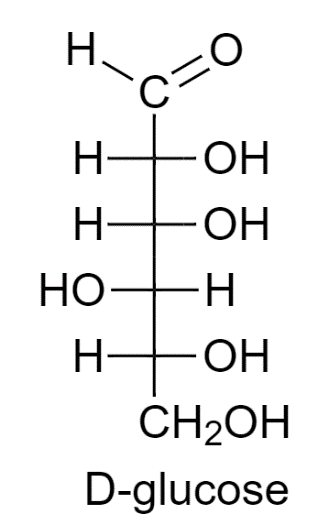
D Glucose Fischer Projection
Ever wondered how scientists keep track of molecules that are mirror images of each other? It's like trying to tell twins apart – tricky, right? That's where the Fischer projection comes in, especially when we're talking about D-Glucose, a sugar that’s incredibly important for, well, everything living!
So, what is a Fischer projection? Think of it as a simplified way of drawing a 3D molecule on a flat piece of paper (or a computer screen!). It's like a map that shows you the arrangement of atoms around a central carbon atom, specifically in carbohydrate molecules. And trust me, carbohydrates get pretty complex. Without a clear, standard way to represent them, things would get confusing fast.
The beauty of the Fischer projection lies in its simplicity. It uses a cross shape: vertical lines represent bonds going behind the page, and horizontal lines represent bonds coming out towards you. It's a shorthand, a secret language that chemists use to quickly understand the structure of sugars like D-Glucose. Imagine trying to describe a complicated building without a blueprint! That's what working with sugars would be like without Fischer projections.
Must Read
Now, let's zoom in on D-Glucose. D-Glucose is a chiral molecule, meaning it exists in two forms that are mirror images of each other (like your left and right hands). These forms are called enantiomers, and they're designated as either 'D' or 'L'. The Fischer projection is essential for distinguishing between these two forms. For D-Glucose, the hydroxyl (OH) group on the bottommost chiral carbon (the one furthest from the aldehyde group) is on the right side of the projection. That's what makes it 'D'! If that OH group were on the left, it would be L-Glucose.
Why is this important? Well, our bodies are incredibly picky! Enzymes, the biological catalysts that drive pretty much all chemical reactions in our bodies, are designed to interact with specific molecules. They're like locks that only a certain key can open. Most enzymes in our bodies are designed to work with D-Glucose, not L-Glucose. So, even though they're mirror images, they have very different biological effects.
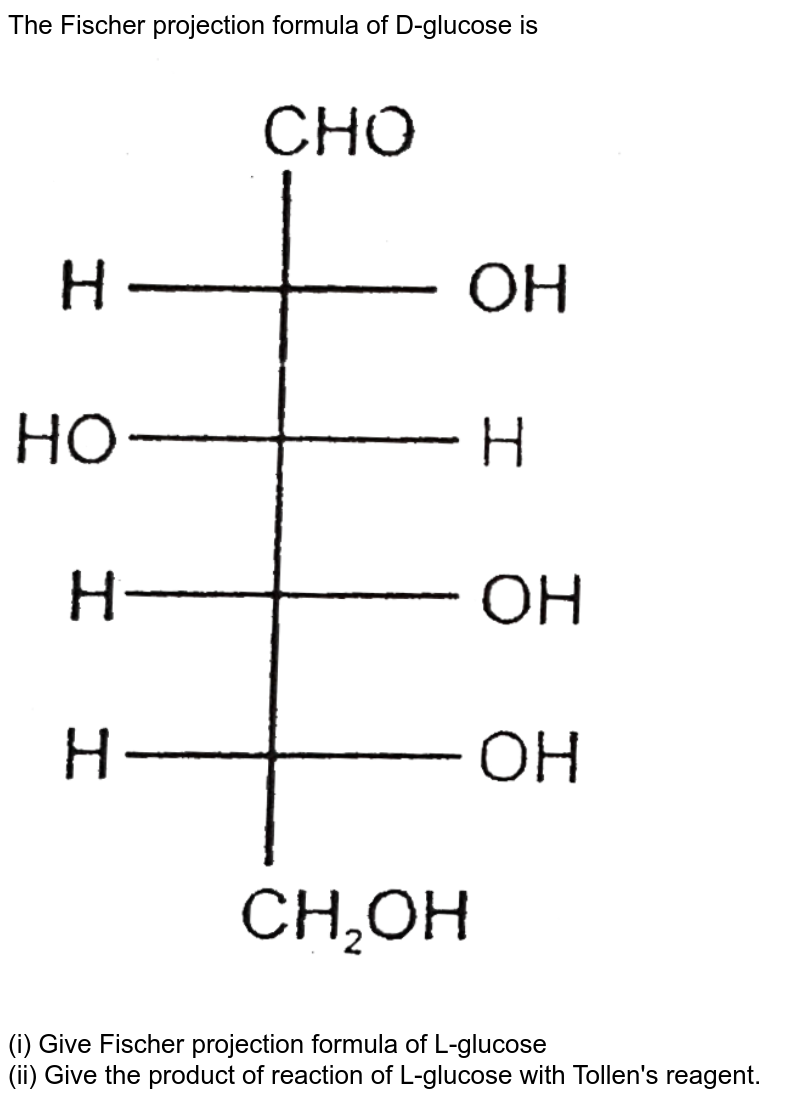
The benefits of using Fischer projections extend beyond just identifying D and L forms of sugars. They also allow us to quickly compare different sugars and understand how their structures relate to their properties. For example, slight changes in the arrangement of OH groups on a glucose molecule can turn it into a different sugar, like galactose or mannose. The Fischer projection lets us easily see those differences and understand how they might affect how the sugar tastes, how it's metabolized, or how it interacts with other molecules.
In short, the D-Glucose Fischer projection is a simple yet powerful tool that helps us understand the complex world of carbohydrates. It allows us to distinguish between mirror images, compare different sugars, and ultimately understand how these molecules interact with our bodies and the world around us. So, next time you hear about sugars, remember the handy-dandy Fischer projection – it’s the secret decoder ring of the carbohydrate universe!