Copper Conducts Electricity Physical Or Chemical

Okay, folks, let's talk about copper! That shiny, reddish-brown metal we see everywhere – in our pennies (well, mostly zinc now, but that's another story!), our wires, and even sometimes glinting on fancy pots and pans. We all know copper conducts electricity, right? But the million-dollar question is: is that a physical or a chemical property?
Now, some science stuff can feel like deciphering ancient hieroglyphics, but I promise, this one's easier than making toast. Seriously! Think about it this way:
Physical vs. Chemical: The Ultimate Showdown!
Imagine you're at a party. (A science party, naturally!) Physical properties are like outward appearances. Think hair color, height, maybe even that sparkly lab coat you're rocking. You can observe them without, you know, blowing anything up or changing what something is at its core.
Must Read
Chemical properties, on the other hand, are all about how things react with each other. It’s like finding out someone’s secret talent for, say, dissolving things in acid (please don’t actually do that!). To see a chemical property, you usually need to do something – mix things, heat things up, maybe even introduce a rogue element (like my Uncle Jerry at Thanksgiving dinner). The substance itself changes!
Copper's Electrifying Talent: A Closer Look
So, let’s put copper under the microscope (figuratively speaking, unless you really want to get out your microscope). Does it change into something else when it conducts electricity? Does it suddenly sprout wings and start singing opera? (Okay, maybe I'm exaggerating... just a tad.)
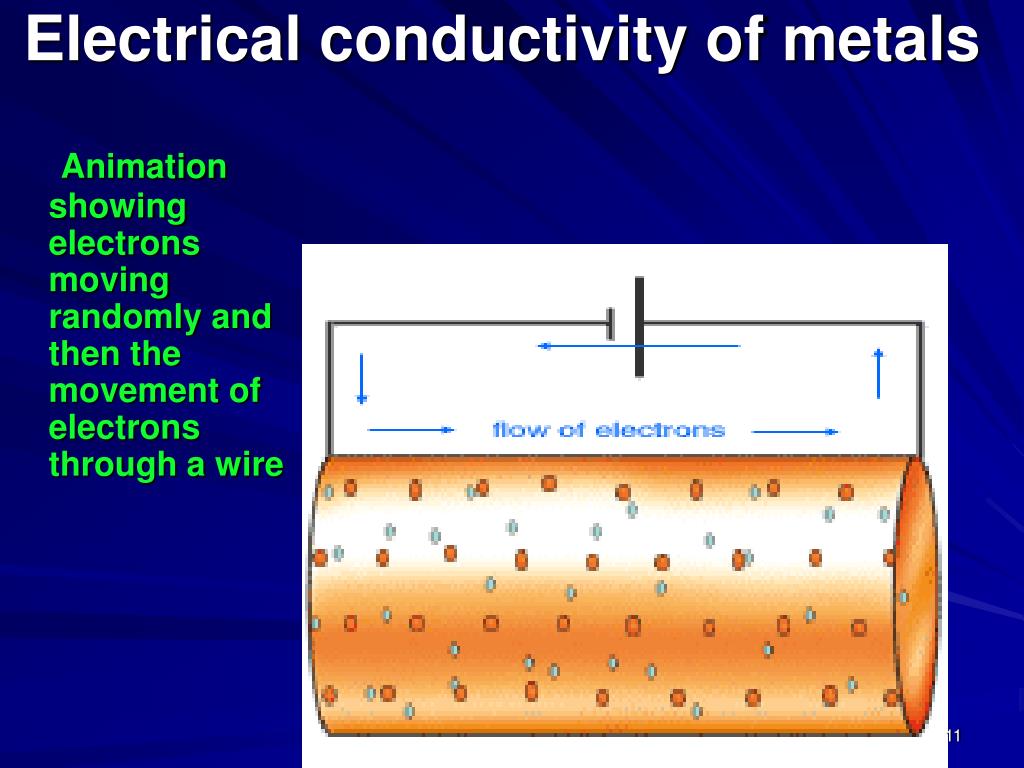
The answer, my friends, is a resounding NO!
When copper conducts electricity, it's basically acting like a super-efficient highway for electrons. Those tiny little particles are zipping through the copper like race cars on the Indy 500. The copper allows them to move, but it doesn't transform into something new. It's still good old copper, just a copper that’s currently hosting a raging electron party.

Think about it: If copper turned into, say, cheese every time electricity flowed through it, our electrical grid would be a cheesy mess! And we'd have a serious rodent problem.
"Conductivity is a physical property because the chemical composition of copper doesn't change when it allows the flow of electrical current." - My (imaginary) Science Guru
Therefore, conducting electricity is a physical property of copper. Huzzah!

But wait! Some of you clever cookies might be thinking, "But what about corrosion? Doesn't copper turn green over time? Isn’t that a chemical change?”
Ah, yes! You’re absolutely right. Copper can corrode, forming that greenish layer called patina. That's a chemical reaction with the oxygen and carbon dioxide in the air (and sometimes pollutants). Copper is reacting and forming a new compound. But that's not the same as simply conducting electricity.

Imagine it like this: Copper can be a friendly neighbor (conducting electricity, a physical property). But it can also get into arguments with the atmosphere and start forming patina (corrosion, a chemical property). It's a multifaceted metal!
So, to recap: Copper conducting electricity is like a perfectly executed dance move – graceful, efficient, and leaves the dancer (the copper) unchanged. It's a physical property. And now, armed with this knowledge, you can confidently impress your friends at your next (hopefully non-cheesy) science party!
You're welcome!
