Compare And Contrast Thermal Energy And Temperature
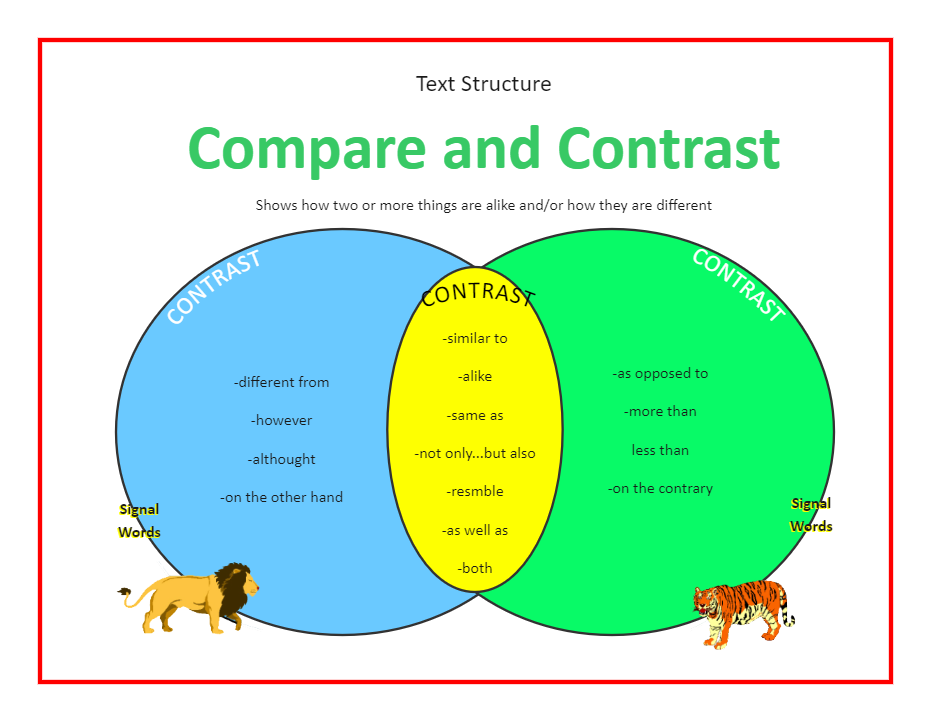
Ever wondered what really makes things hot or cold? It's a tale of two concepts: Thermal Energy and Temperature. They might sound like twins, but trust me, they're more like quirky cousins!
Let's dive into this surprisingly fun comparison.
What’s the Deal with Temperature?
Think of temperature as the average speed of tiny, invisible particles. These particles are always jiggling around! Temperature basically tells you how fast they're moving on average.
Must Read
A high temperature means they're zooming. A low temperature? They're practically doing the slow dance. Simple, right?
We usually measure temperature with good old tools like thermometers. Celsius, Fahrenheit, Kelvin... they're all just different ways to measure the average jiggle.
Okay, So What's Thermal Energy Then?
Thermal Energy is the total energy of all those moving particles. It's not just about the average; it's about absolutely everyone! This includes all the kinetic and potential energy of the particles.
Think of it like this: a swimming pool and a teacup can both be the same temperature. But the pool has WAY more water... and therefore WAY more total energy bouncing around.
That pool has a lot more thermal energy ready to do something cool... like warm you up on a summer day.
The Hilarious Differences: Size Matters!
Here's where things get interesting. Imagine a tiny sparkler and a giant bonfire. They both might burn at roughly the same temperature.
But whoa, that bonfire has a lot more thermal energy! It's got way more fuel, more particles moving, and more potential to roast marshmallows.
Temperature is just one measurement. Thermal energy is the grand total, the whole enchilada!
Imagine This: Ice, Ice, Maybe?
Let’s say you have an iceberg, and a single ice cube. Both are zero degrees Celsius (32 Fahrenheit). That's their temperature.
But the iceberg contains tremendously more thermal energy than that ice cube. It could cool down a lot more drinks, if you had the right glass!
It's because the iceberg has way, way more ice molecules doing the temperature thing. And the "doing" takes the form of thermal energy.
Why Should You Even Care?
Understanding this difference is super helpful in all sorts of scenarios. Think about cooking: You're not just worried about the oven's temperature.
You also care about how much thermal energy the food can absorb before it's perfectly cooked. That's why a small potato cooks faster than a giant turkey!
Or consider engines. They convert fuel into thermal energy, which then gets turned into motion. The more efficient they are, the better they convert that energy.

Transfer of Energy: Hot Potato, Anyone?
Heat, that thing we often hear about, is simply the transfer of thermal energy from one thing to another. Imagine touching a hot stove (don't actually do it!).
Thermal energy flows from the hot stove to your hand. Ouch! That heat transfer keeps going until both things reach the same temperature.
This process is called reaching thermal equilibrium. It's all about the balance of energy. Mother Nature's constantly balancing the books. And believe me, she's got a lot of energy to balance.
Cool Examples in Action!
Think about a blacksmith forging metal. The blacksmith heats the metal to a high temperature to make it malleable.
But it's the high thermal energy that allows them to bend and shape the metal. The metal has absorbed enough energy to change its form.
The blacksmith is essentially manipulating thermal energy to create something new. Talk about a hot job!
Insulation: Keeping the Heat Where You Want It
Insulation works by slowing down the transfer of thermal energy. A thermos keeps your coffee hot by preventing heat from escaping.

It doesn't change the coffee's temperature (at least, not right away). It just keeps all that lovely thermal energy bottled up inside.
It's like a force field protecting your caffeine fix. And thank goodness for that!
The Takeaway: It's All Relative
Thermal Energy is the total energy. Temperature is the average. Think of it this way: a swarm of bees vs. a single bee.
The swarm as a whole has more buzzing energy. And, even if both swarm and single bee move at the same speed. But each bee's speed can be considered to be the temperature in this scenario.
They're related, but they tell you different things about what's going on. This seemingly small difference has big implications in many area.
Beyond Hot and Cold: What Else is Interesting?
The relationship between thermal energy and temperature gets even weirder when you start talking about phase changes. Like when ice melts into water.
You can pump heat into ice at 0 degrees Celsius, and its temperature won't change until it's all melted. All that energy is going into changing the state of the water, not raising its temperature.
Mind. Blown. Nature, you crazy!
So, Are You Hooked Yet?
Hopefully, this has sparked your curiosity about the amazing world of heat and energy. Thermal energy and temperature are fundamental concepts.
They are very applicable to everything from cooking to climate science to the design of super-fast computers.
Don't be afraid to explore further! There's a whole universe of fascinating science waiting to be discovered. And this is just the tip of the iceberg.
Final Thoughts: A Dynamic Duo
So, the next time you're enjoying a hot cup of coffee or shivering on a cold day, remember the dynamic duo of thermal energy and temperature.
They're working hard behind the scenes, making the world around us tick. And with a little understanding, you can appreciate the science behind it all.
And that’s pretty cool! Now, go forth and impress your friends with your newfound knowledge.
