Co3 2- Molecular Geometry

Okay, so you've probably never sat down at a dinner party and casually brought up "Co3 2- molecular geometry," right? Unless you're hanging out with a seriously cool bunch of chemists. But stick with me, because this isn't as scary as it sounds. Think of it like arranging furniture, but, you know, with way tinier things.
What even is Co3 2-?
First, let's break it down. Co3 2- is a polyatomic ion called the carbonate ion. It's basically one carbon atom (C) hanging out with three oxygen atoms (O3), and they've got a little extra negative charge to throw around (2-). Imagine it like a group of friends who are always a little bit extra...in a good way, of course!
Now, these atoms don't just huddle together randomly. They arrange themselves in a very specific way, like teenagers trying to figure out the perfect group selfie. This arrangement is what we call its molecular geometry.
Must Read
Trigonal Planar: The Party Planner of Geometries
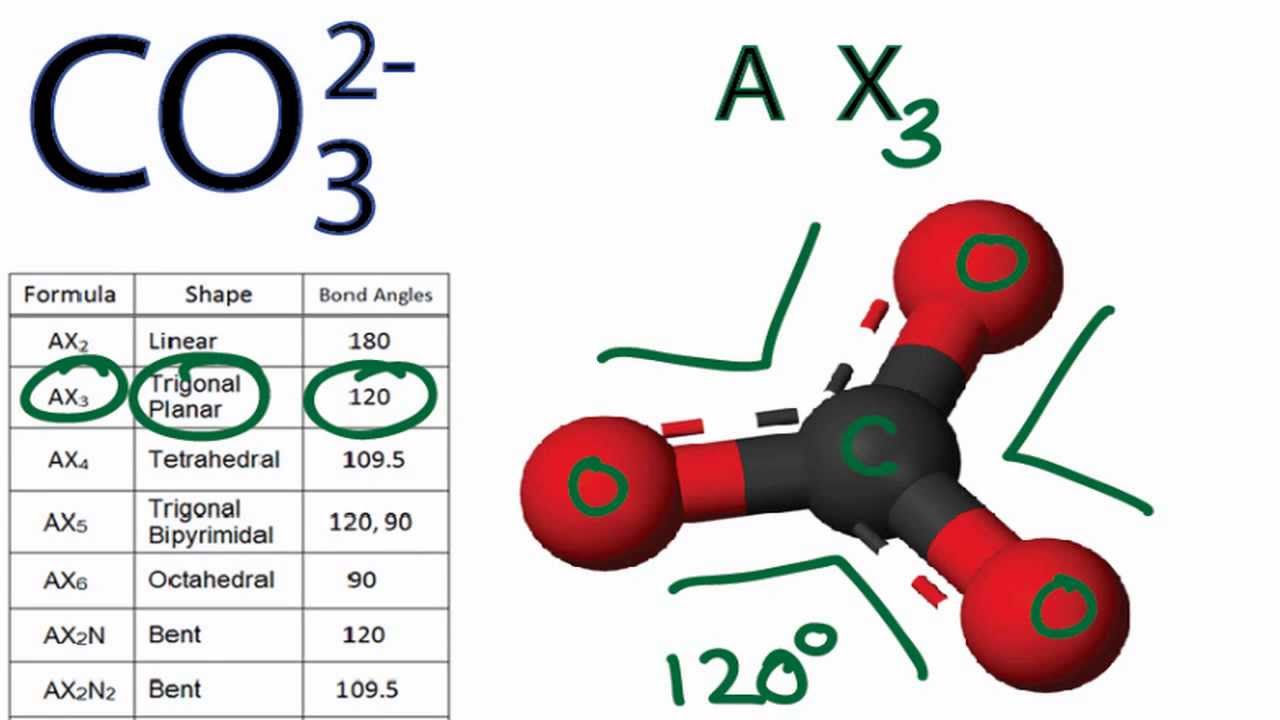
The carbonate ion's molecular geometry is called trigonal planar. Say that five times fast! Basically, it means the carbon atom sits in the middle, and the three oxygen atoms are equally spaced around it, all on the same flat plane. Think of it like a perfectly symmetrical pizza with three toppings evenly spaced around it. No topping gets more love than the others. #EqualityForToppings
Imagine you're planning a party (again, cooler than your average dinner party). You have one central table (the carbon atom), and you want to place three chairs (the oxygen atoms) around it. To give everyone the same amount of personal space and make the whole setup look aesthetically pleasing, you'd arrange the chairs in a triangle, right? That's essentially what's happening with the carbonate ion. It's all about minimizing repulsion between those negatively charged oxygen atoms.

Bond angles are super important in molecular geometry. The bond angle in a trigonal planar molecule is 120 degrees. What that means is that if you were to measure the angle from one oxygen atom to the carbon atom in the middle, and then to another oxygen atom, the angle would be 120 degrees. That's how we know they're evenly spaced!
Why does this even matter?
Okay, so why should you care if the carbonate ion is shaped like a pizza or a lopsided starfish? Well, molecular geometry dictates a molecule's properties. It's the "why" behind how it interacts with other molecules, dissolves in water, or even tastes (though please don't go around tasting carbonate ions!).
For instance, carbonate ions are crucial for maintaining pH balance in our blood and oceans. They're also a key component of limestone and other rocks. Think of them as tiny, geometric building blocks that make up the world around us. Not bad for a bunch of atoms having an existential crisis.
Lone Pairs: The Wallflowers of Molecular Geometry
Now, sometimes molecules have extra electrons that aren't involved in bonding, called lone pairs. These lone pairs act like awkward wallflowers at the party, taking up space and influencing the shape of the molecule. Luckily, the carbonate ion doesn't have any lone pairs hanging around on the central carbon atom. That's why it's a nice, neat trigonal planar shape!
So, the next time you're enjoying a slice of pizza (preferably one with equally spaced toppings) or marveling at a limestone cliff, remember the carbonate ion and its perfectly arranged atoms. It might just make you appreciate the molecular world a little bit more. And hey, you'll have some seriously impressive trivia to drop at your next party. Just don't be surprised if people look at you funny. Unless, of course, it's a chemistry party. Then you'll be the star!


