Co Molecular Orbital Diagram
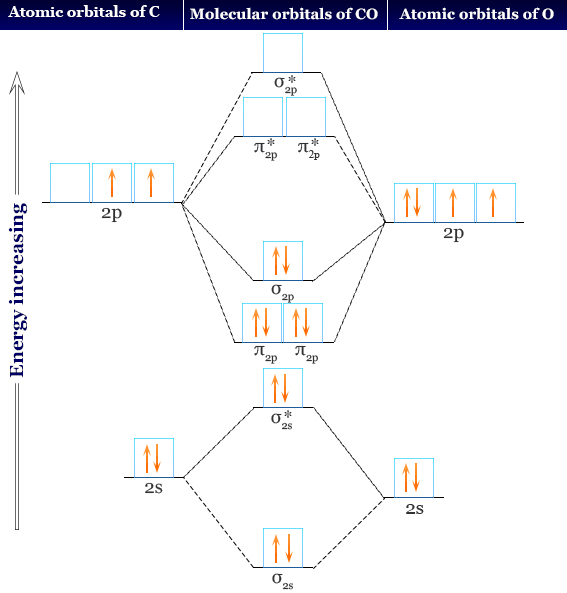
Hey there, molecule lover! Ever heard of a CO Molecular Orbital Diagram? Sounds intimidating, right? Nah! It's actually kinda cool. Think of it as a blueprint for how carbon and oxygen cuddle up to form carbon monoxide (CO).
What’s the Big Deal About CO?
Okay, CO isn't exactly known for being cuddly in real life. It's a poisonous gas! But stick with me! Understanding its electronic structure is like understanding the secret to its, well, not-so-secret deadliness. So, why does this molecule matter?
Think about car exhaust. Think about incomplete combustion. CO is everywhere, and understanding it helps us build better catalytic converters, safer engines, and maybe even figure out how to trap it and turn it into something useful! Who knows?
Must Read
Diving into the Diagram: It’s Easier Than You Think!
Alright, let's get down to the nitty-gritty. A molecular orbital diagram is basically a way of showing how the atomic orbitals (those fuzzy clouds where electrons live around individual atoms) combine to form molecular orbitals when atoms bond.
Imagine two kids (carbon and oxygen) playing with LEGO bricks (their electrons). They each have their own pile of bricks (atomic orbitals). When they build something together (form CO), they share the bricks and make something new (molecular orbitals). Some LEGO bricks combine constructively (bonding orbitals), making the structure stronger. Others combine destructively (antibonding orbitals), weakening it.
It's like a microscopic tug-of-war between attraction and repulsion! And the molecular orbital diagram? That’s the instruction manual for their LEGO creation!
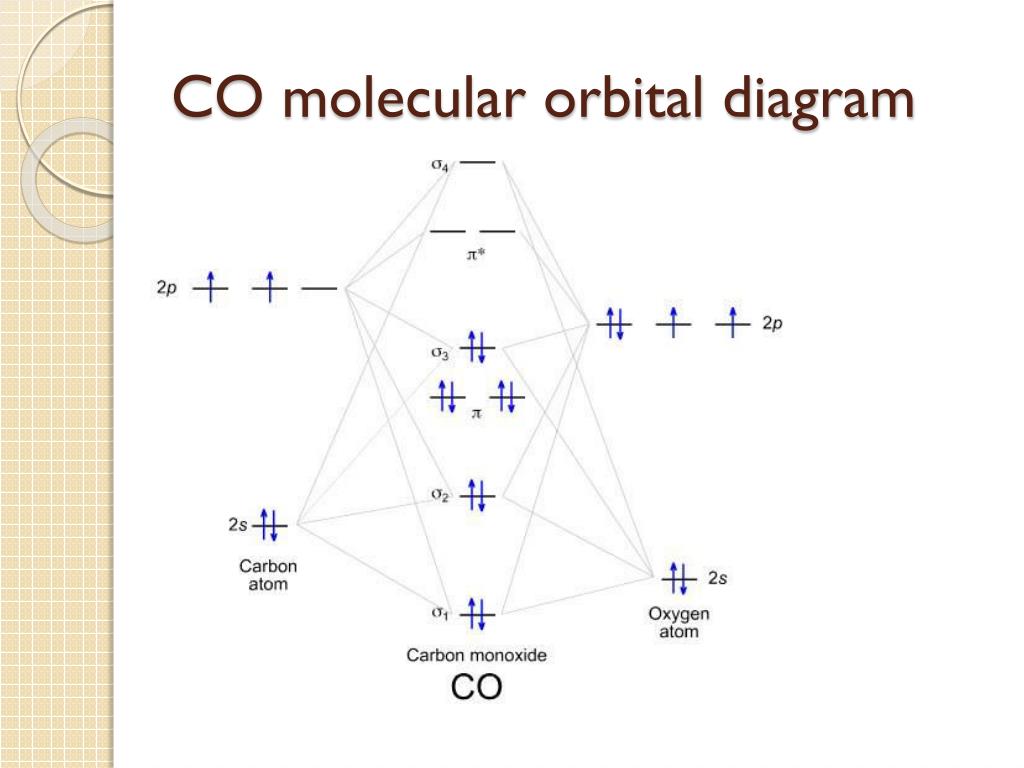
The Players: Sigma and Pi
Now, a quick vocab lesson. You'll hear about sigma (σ) and pi (π) orbitals. Sigma orbitals are formed by head-on overlap of atomic orbitals. Think of it as a direct handshake. Pi orbitals are formed by sideways overlap. More like a high-five.
CO's diagram has both sigma and pi orbitals. We’ve got sigma bonding orbitals (super stable!), sigma antibonding orbitals (not so stable!), pi bonding orbitals (also stable!), and pi antibonding orbitals (you guessed it: unstable!).
Fun Fact: The asterisk symbol () usually denotes an antibonding orbital. So, σ is a bad guy! (electronically speaking).
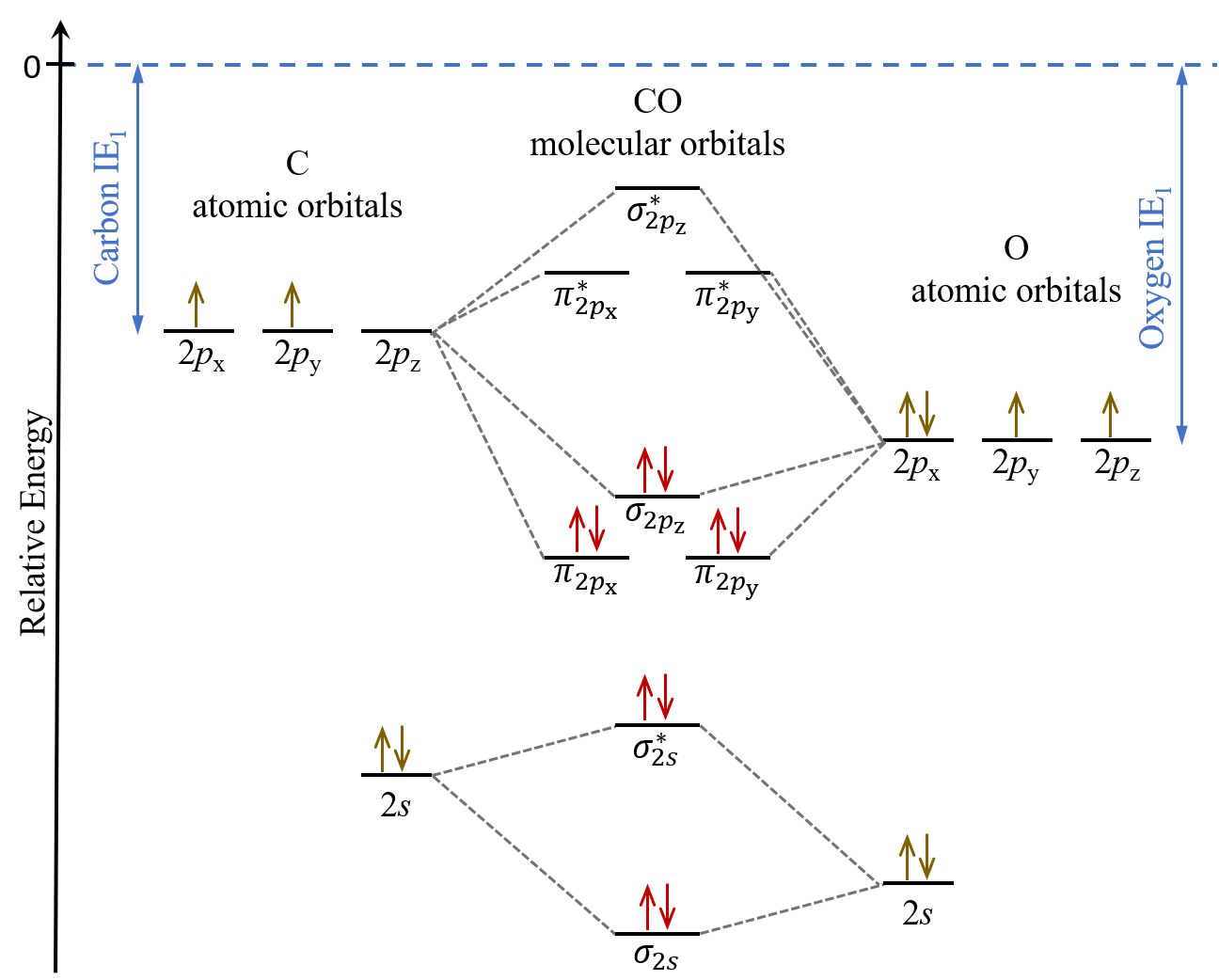
Filling Up the Orbitals: The Electron Party
Carbon has 4 valence electrons. Oxygen has 6. That's a grand total of 10 electrons ready to party! The Molecular Orbital Diagram is the dance floor. But electrons aren't party animals; they follow rules! They fill the lowest energy orbitals first (think of it like grabbing the best snacks on the buffet table). Each orbital can only hold two electrons (Pauli Exclusion Principle, the bouncer at the door!).
So, we start filling the sigma bonding orbital, then the sigma antibonding orbital, then the pi bonding orbitals, and so on. Once we know how all the orbitals are filled, we can figure out the bond order.
Bond Order: How Much Love is There?
The bond order tells us how many bonds are between the atoms. It’s a measure of the strength of the attraction. The formula is simple:

(Number of electrons in bonding orbitals – Number of electrons in antibonding orbitals) / 2
For CO, it's (8 – 2) / 2 = 3. That means CO has a triple bond! Pretty darn strong, huh? That's why it takes a lot of energy to break it apart. Strong attraction is the way to go!
Quirky CO: It's a Little Backwards
Here's where things get really fun (and slightly weird). The molecular orbital diagram of CO is… different. It’s not exactly what you'd expect based on the electronegativity difference between carbon and oxygen. The highest occupied molecular orbital (HOMO) – the last orbital to get filled – is actually slightly more concentrated on the carbon atom than you might think!

This means that CO can sometimes bond to metals through the carbon end! Crazy, right? Oxygen is usually the one doing the electron hogging, but in CO, carbon is a little more eager to share... or at least, more readily available for bonding.
Why Bother? It's All About Understanding
So, why should you care about this crazy diagram? Because it helps us understand how CO interacts with other molecules, how it absorbs light, and how it acts as a ligand (a molecule that binds to a metal). It’s a window into the quantum world, explaining how atoms connect and form the molecules that make up everything around us.
It’s also a reminder that even something as seemingly simple as carbon monoxide has hidden complexities and fascinating secrets waiting to be discovered. Pretty neat, huh?
Now go forth and impress your friends with your newfound knowledge of CO molecular orbital diagrams! (Or at least, don't bore them too much.)
