Chromium Ii Phosphate Net Ionic Equation
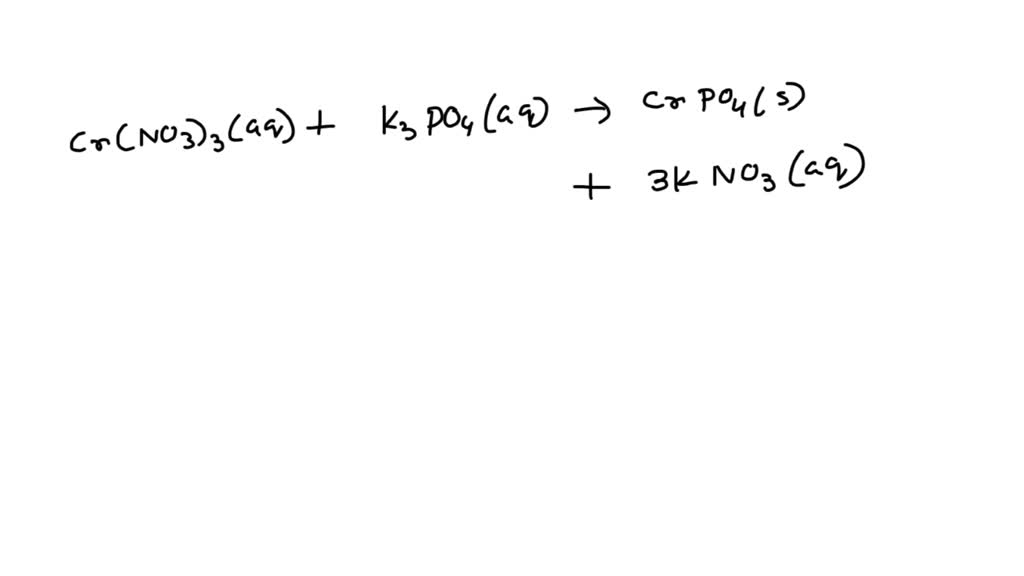
Ready for some chemical equation fun? Let’s dive into the world of Chromium(II) Phosphate! Don't worry, it's not as scary as it sounds. Think of it as a super-simplified story of what happens when certain chemicals get together and decide to mingle in water.
We're talking about the net ionic equation, the bare-bones version of a chemical reaction. It's like the director's cut – all the fluff is removed, leaving just the action-packed scenes with the main characters. In this case, our main characters are the ions directly involved in forming a solid (a precipitate). We are only showing the ingredients that are active in the reactions.
Imagine you're planning a party. A regular chemical equation would list everything you bought: the cake, the balloons, the plates, even the ice. But the net ionic equation? It only shows the ingredients that combine to actually create the cake! Everything else gets filtered out. That's why it's so cool – it's all about efficiency!
Must Read
So, What's the Big Deal with Chromium(II) Phosphate?
Chromium(II) phosphate, or Cr3(PO4)2, is like that shy kid at the party. It doesn't exactly dissolve readily in water. Instead, it forms a solid. This "solid-forming" action is the key. It is the star of the show in the net ionic equation.
To get to the net ionic equation, we have to think about what's actually happening with the reactants. Typically, you'd start with solutions containing chromium(II) ions (Cr2+) and phosphate ions (PO43-). Think of them as swimming around freely in the water, looking for partners.
When these two ions find each other, BAM! A bond forms. The Cr2+ and PO43- ions get together and create our solid, Chromium(II) Phosphate. All the other spectator ions - the ions that were there but didn't change, they are not represented.
Let's picture the net ionic equation:

3Cr2+(aq) + 2PO43-(aq) → Cr3(PO4)2(s)
Isn't it elegant? It shows exactly what's reacting, where the state of the substances (aqueous or solid), and how everything comes together. No unnecessary details, just pure chemical reaction bliss.
Why is This Entertaining?
Okay, maybe "entertaining" is a strong word for everyone. But for chemistry enthusiasts (and those about to become enthusiasts!), the net ionic equation is like a magic trick. It takes a seemingly complex reaction and boils it down to its essential components.
It's like understanding the punchline of a joke. Once you "get" the net ionic equation, you understand the driving force behind the reaction. You can predict whether a precipitate will form, and that's strangely satisfying. It lets you play detective and figure out what will happen before it happens.

Furthermore, the net ionic equation helps you to understand reactions better. Not only do they help to predict reaction results but they also provide insights into the way reactions occur in a solution.
It's also a really good excuse to draw colorful ions bonding together. You could make it into a comic book! Chromium(II) ions, the misunderstood heroes, finally finding their phosphate partners and settling down as a stable, solid compound. Okay, maybe that's just me…

Give it a Try!
Want to experience the thrill of simplifying chemical reactions yourself? Look up other precipitation reactions and try to write their net ionic equations. You'll be amazed at how much easier it is to understand what's happening when you strip away the excess baggage.
You might even find yourself muttering "Cr3(PO4)2(s)" under your breath. Don't worry; we won't judge. Welcome to the exciting world of net ionic equations!
So go forth, experiment, and have fun uncovering the simplified truth behind complex reactions. Chemistry is waiting. Good Luck!
