Chlorine Pentafluoride Molar Mass

Okay, let's talk about Chlorine Pentafluoride, but don't worry, we're not diving into a super-complicated chemistry lecture. Think of this as more of a friendly chat about something you might vaguely remember from high school chemistry… or not! We’re going to explore its molar mass. But first, let's break down what that even means in a way that makes sense, even if you haven't seen a beaker in years.
So, what's a molar mass? Imagine you're baking cookies. A recipe calls for "one cup" of flour. But what exactly does that mean? Different flours have slightly different weights for the same volume (a cup). Molar mass is like a super-precise, universal "cup" for elements and compounds in the chemistry world. It's the mass of one mole of a substance. Think of a "mole" like a dozen – it's just a specific number (a REALLY big number, actually, 6.022 x 1023, but don't worry about memorizing it!). Basically, molar mass tells you how much a specific number of molecules of something weighs.
Why should you even care about the molar mass of Chlorine Pentafluoride (ClF5)? Fair question! While you probably won't be using it in your kitchen (unless you’re a very adventurous… and highly trained… chemist!), understanding molar mass is fundamental to understanding chemistry. It’s like knowing the notes on a musical scale – you need to know them to understand and appreciate the music. In this case, understanding molar mass lets us predict how ClF5 will react, how much we need for a reaction, and generally understand its properties. Imagine it like needing the right amount of yeast to make bread rise properly. Too much, and you've got a disaster. Too little, and you've got a flat loaf. Chemistry is all about these precise ratios!
Must Read
Breaking Down ClF5
Now, let's tackle ClF5 itself. As the name suggests, it's made up of one chlorine atom (Cl) and five fluorine atoms (F). To find its molar mass, we need to look at the periodic table. The periodic table is like the chemist's bible – it tells you everything about the elements, including their average atomic masses. These atomic masses are measured in atomic mass units (amu), which are so tiny that we use grams per mole (g/mol) when we're working with real-world quantities.
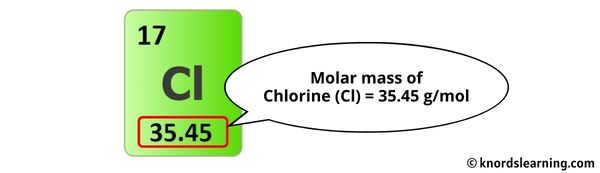
On the periodic table, you'll find that chlorine (Cl) has an atomic mass of approximately 35.45 g/mol. Fluorine (F) has an atomic mass of approximately 19.00 g/mol. These are just averages based on the naturally occurring isotopes (different versions) of each element.

Calculating the Molar Mass
Okay, here's where the math comes in, but it's super simple! Since we have one chlorine atom and five fluorine atoms in ClF5, we just need to add up their individual masses. It's like calculating the total cost of a shopping trip. One chlorine is like buying one apple and five fluorines are like buying five oranges.
So, here’s the formula:
Molar Mass of ClF5 = (1 x Molar Mass of Cl) + (5 x Molar Mass of F)
Molar Mass of ClF5 = (1 x 35.45 g/mol) + (5 x 19.00 g/mol)
Molar Mass of ClF5 = 35.45 g/mol + 95.00 g/mol
Molar Mass of ClF5 = 130.45 g/mol

Therefore, the molar mass of Chlorine Pentafluoride (ClF5) is approximately 130.45 g/mol. That means one mole (that giant handful!) of ClF5 weighs about 130.45 grams.
Why does this matter? Well, let's say you were trying to perform a reaction involving ClF5. Knowing its molar mass would allow you to accurately measure out the correct amount to react with other chemicals. It’s like following a recipe precisely to get the perfect cake.
Although ClF5 is not something you'll encounter daily, the principles of molar mass are crucial in countless scientific and industrial applications. From drug development to materials science, knowing how much of something you have is essential for everything.

Real-World Relevance (Even if it Seems Obscure!)
You might be thinking, "Okay, that's neat… but still, I’m never going to use this." And that’s probably true! But consider this: understanding molar mass is a building block for understanding how the world works at a molecular level. It’s like learning basic arithmetic – you might not be crunching numbers every day, but it helps you understand concepts like budgeting, cooking, and even understanding sports statistics.
Think about making a specific concentration of cleaning solution. You need to know the molar mass of the active ingredient to dilute it properly. Or consider the dosage of a medicine; understanding molar mass helps ensure the right amount of the active pharmaceutical ingredient is delivered. These are everyday applications that stem from the same fundamental concept!
So, even though ClF5 itself might seem like an obscure chemical, understanding its molar mass, and molar mass in general, is a window into understanding the world around us a little bit better. It's a fundamental concept in chemistry that underlies countless applications, from making sure your cookies bake just right to developing life-saving medications. And hopefully, now, it feels a little less intimidating and a little more… interesting!
