Chlorine Lewis Dot Structure

Okay, picture this: you're at a party. A chemistry party, naturally (because who needs awkward small talk when you can talk about electron configurations?!). Everyone's mingling, trying to find someone to bond with. And there's Chlorine, standing in the corner, looking…well, a little dotty.
Chlorine: The Lone Wolf of the Periodic Table
Chlorine (Cl) is element number 17. It's that yellowish-green gas that makes swimming pools smell, well, like swimming pools. But forget the smell for a second. Focus on its atomic structure. It's got 17 protons, 17 neutrons (ish, depends on the isotope, but let's not get bogged down in that detail right now), and, crucially, 17 electrons zooming around it.
Now, these electrons are arranged in shells. Think of them like stadium seating around a tiny atomic football field. The first shell can hold 2 electrons, the second can hold 8. So far, so good. That leaves us with… 7 electrons! These are our valence electrons, the cool kids who hang out on the outermost shell and decide how Chlorine interacts with the world.
Must Read
Enter the Lewis Dot Structure: The Social Media Profile for Atoms
This is where the Lewis Dot Structure comes in. It’s basically a super simplified, atomic dating profile. Instead of a photo of your best side, it shows how many valence electrons an atom has. We write the element symbol (Cl, duh!), and then we draw dots around it to represent those 7 valence electrons.
Imagine Chlorine wearing a cool, slightly asymmetrical crown of seven electron-jewels. These electron-jewels aren't just randomly placed; they follow a specific rule. We pretend there are four spots around the Chlorine symbol (top, bottom, left, right). We fill each spot with one electron until we've used up all four, then we start pairing them up. Like a lonely hearts club where atoms are trying to find their perfect match.
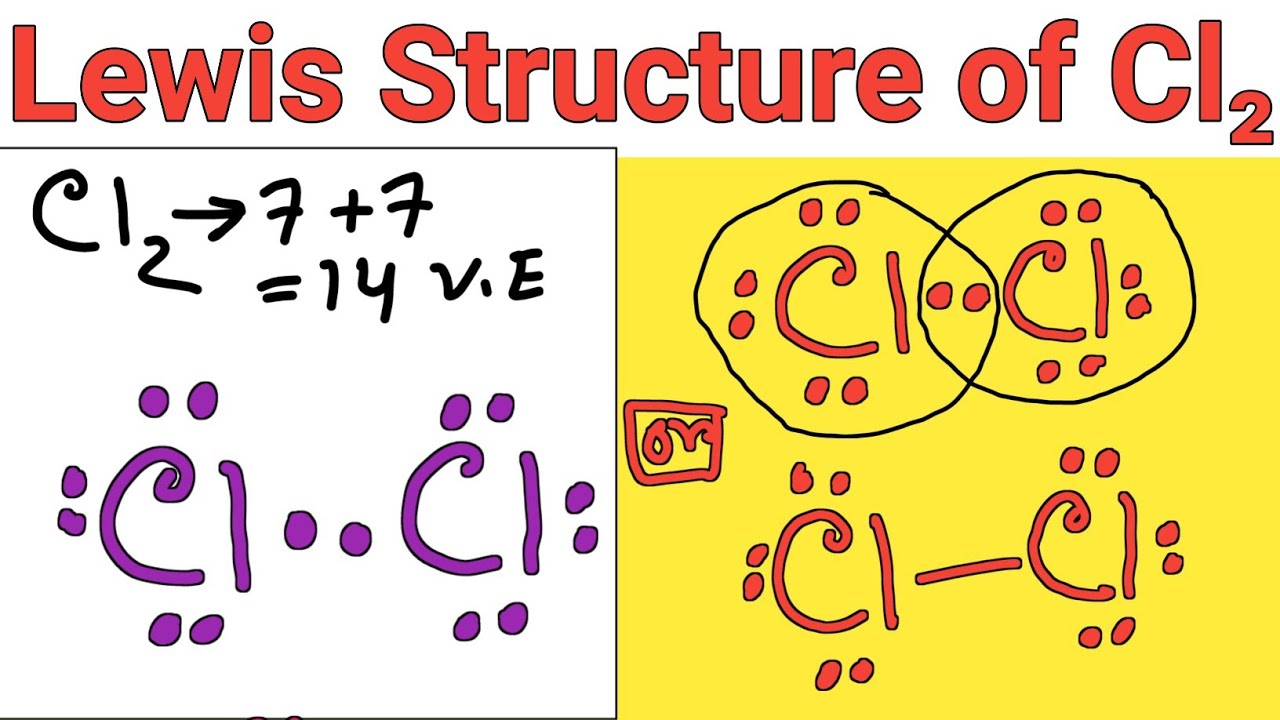
So, Chlorine's Lewis Dot Structure looks something like this (if I could draw it here): Cl with two dots on top, two dots on the bottom, two dots on the left, and ONE lonely dot on the right. That’s it! Simple, right?
That lonely electron is begging for a partner. It's Chlorine's "desperately seeking" signal. It screams, "I need one more electron to complete my octet!" (An octet is the magic number of 8 electrons in the outermost shell that makes an atom stable and happy, like finding the perfect Netflix series to binge-watch).

Chlorine, with its 7 valence electrons, is incredibly reactive. It loves to grab that one extra electron from somewhere, anywhere. This is why it’s used in disinfectants. It's basically stealing electrons from bacteria and viruses, disrupting their structures and rendering them harmless (or, you know, dead).
Why This Matters: The Science Behind the Spark
Knowing the Lewis Dot Structure tells us a lot about how Chlorine will interact with other atoms. It explains why Chlorine forms strong bonds with elements like sodium (Na) to make sodium chloride (NaCl), good old table salt. Sodium, with only one valence electron, is practically throwing itself at Chlorine, shouting, "Take my electron! Complete your octet! Let's be stable together!"

This transfer of an electron from sodium to chlorine creates an ionic bond, a strong electrostatic attraction between the positively charged sodium ion (Na+) and the negatively charged chloride ion (Cl-). It’s like the world's most enthusiastic handshake, but with electrons.
Understanding the Lewis Dot Structure for Chlorine unlocks the secret to understanding its reactivity, its bonding behavior, and its role in countless chemical reactions. From disinfecting your pool to creating life-saving medicines, Chlorine's eagerness to gain that one extra electron makes it an incredibly important element.
So, next time you're at that chemistry party, you can walk right up to Chlorine and say, "Hey, I know your Lewis Dot Structure! You're looking a little electron-deficient; need someone to share with?" You'll be the life of the party, guaranteed. (Okay, maybe not guaranteed. But you'll definitely impress at least one chemist).

