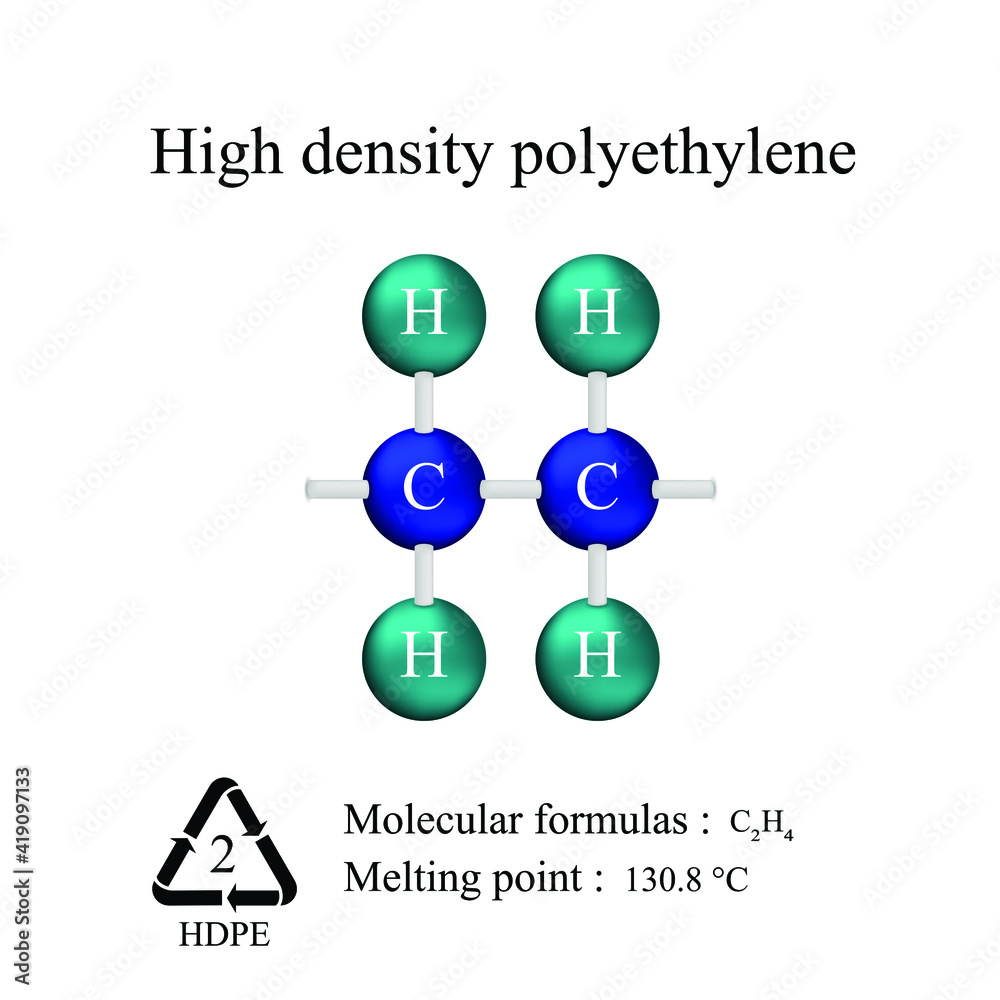
Chemical Formula For High Density Polyethylene
Okay, so picture this: I'm at a picnic, right? Classic summer vibes. I'm about to take a huge bite of my perfectly assembled sandwich when BAM! The plastic picnic table leg decides to give up on life. It just…snapped. Leaving my sandwich inches from dirt-dom. My initial thought? "Ugh, cheap plastic!" But then, because I'm a total nerd (don’t judge!), my brain went, "Wait a minute… what is that plastic actually made of?"
That, my friends, is the story of how I rediscovered my fascination with polymers. And specifically, today we're diving headfirst into the glorious world of High-Density Polyethylene. Or, as the cool kids call it, HDPE.
So, about that plastic picnic table. HDPE is everywhere. Seriously. From milk jugs to detergent bottles, hard hats to those aforementioned picnic tables that clearly need to up their game, HDPE is the workhorse of the plastic world.
Must Read
The Chemical Formula: A Sneak Peek
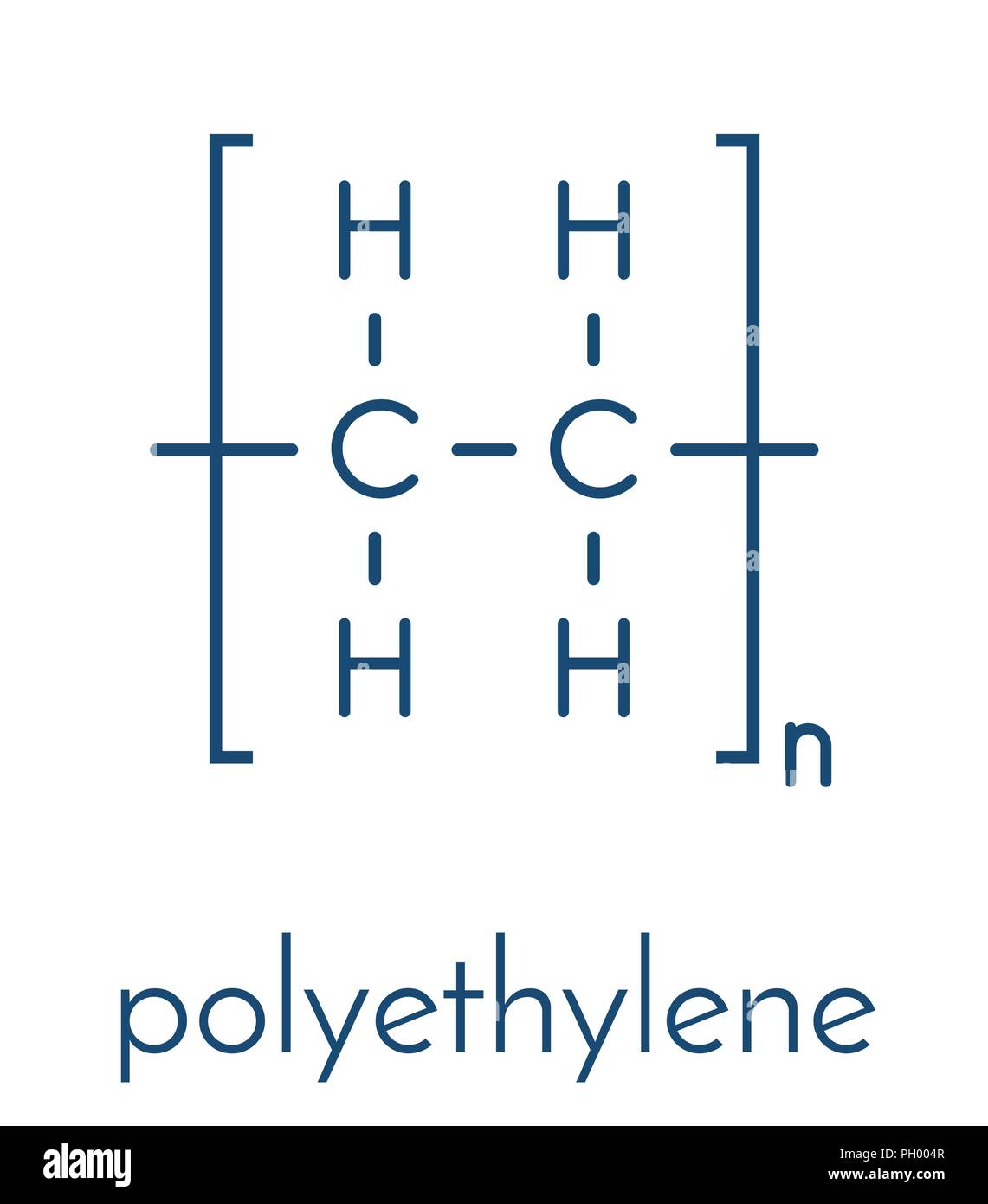
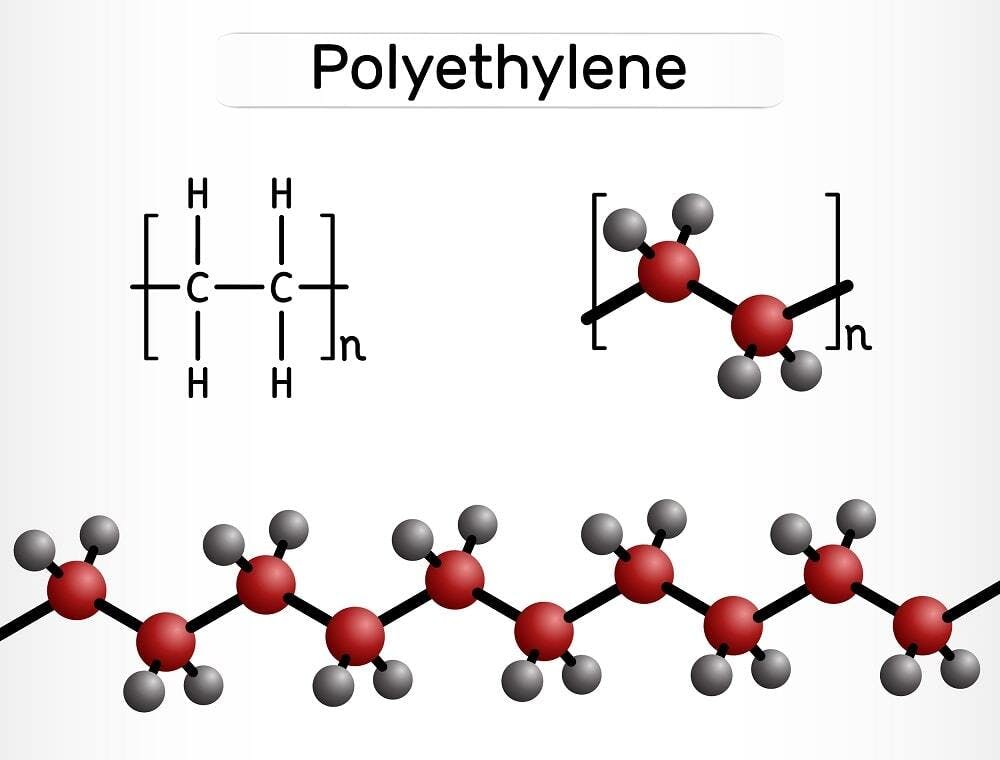
Let's get straight to the point: The chemical formula for HDPE is (C₂H₄)ₙ. Boom. Done. Article over. Just kidding! There's a lot more to unpack here. And trust me, it’s way more interesting than it sounds.
See, that little (C₂H₄) bit is called the monomer. Think of it as a single Lego brick. It's ethylene, which is a simple molecule made of two carbon atoms (C) and four hydrogen atoms (H). Now, the ₙ means "a whole bunch." Like, a seriously whole bunch.
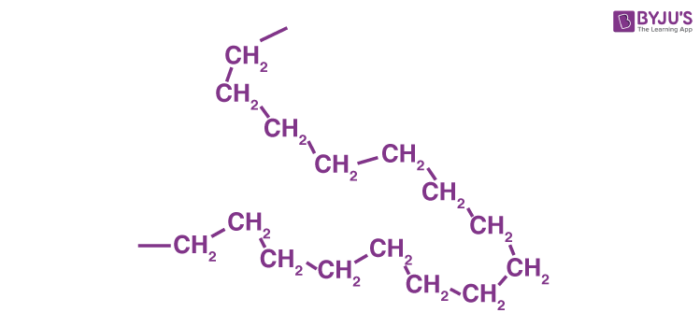
In HDPE, these ethylene monomers link together, forming a long chain. Imagine a massive, never-ending snake made of those Lego bricks. That long chain is the polymer. "Poly" means "many," and "mer" means "unit." So, "many units." Get it? I knew you would!
Side note: Isn't it wild how something as simple as linking together these tiny molecules can create such strong and durable materials? Mind. Blown.
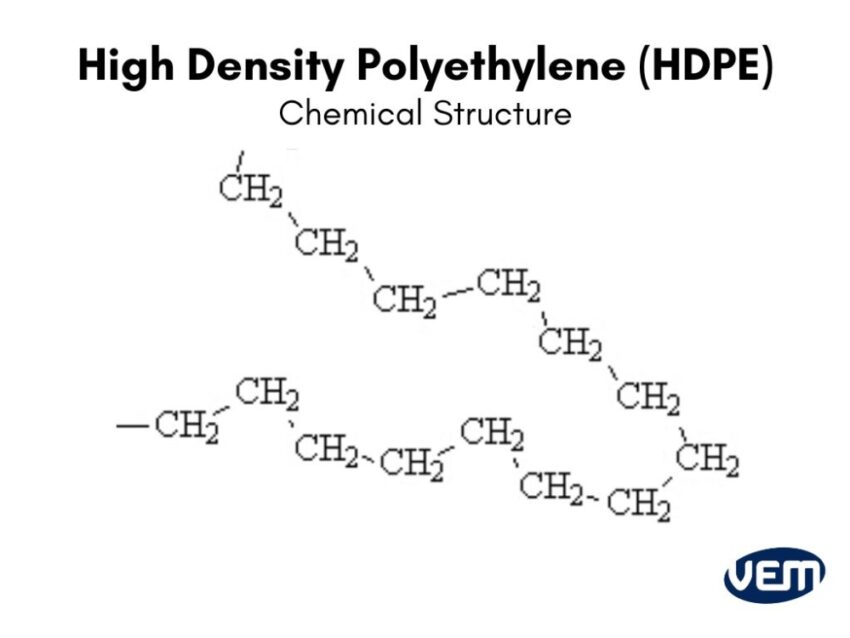
High Density? What's the Deal?
Okay, so we know it’s polyethylene. But what makes it high density? It all comes down to how those long chains pack together.
In HDPE, the chains are relatively straight and have very few branches. This allows them to pack together really tightly. Think of it like neatly stacking uncooked spaghetti noodles in a box. They fit snugly and take up minimal space. Because they're so tightly packed, the material is more dense (hence the name!) and stronger. And, of course, more likely to betray you mid-picnic…
Compare this to Low-Density Polyethylene (LDPE), which has lots of branches along the chain. These branches prevent the chains from packing as tightly. So, it's less dense, more flexible, and typically used for things like plastic bags and squeeze bottles. Basically, less… robust.
Why Should I Care About (C₂H₄)ₙ?
Now, you might be thinking, "Okay, great. I know the chemical formula for HDPE. How is this going to help me in my daily life?" Valid question!
Well, for starters, understanding the basic chemistry of materials allows you to make more informed decisions about the products you use. You can start looking at recycling codes (HDPE is often labeled with a "2" inside the recycling symbol) and think about the properties of the plastic before chucking it in the bin (or, even better, finding a way to reuse it!).
Plus, knowing the chemical formula kind of makes you sound smart at parties. "Oh, this plastic cup? Yeah, that's good ol' (C₂H₄)ₙ at work. Very durable, you know." Okay, maybe not that party-friendly, but you get the idea.
Just remember, with great plastic knowledge comes great plastic responsibility. Recycle, reuse, reduce! The planet will thank you.
Ultimately, even something as seemingly simple as a plastic picnic table leg can lead to a fascinating exploration of the world around us. And who knows, maybe next time you're at a picnic, you'll be able to impress everyone with your knowledge of HDPE. Just try not to let the table collapse on your sandwich. That's never a good look.
