Carbonate Ion Lewis Structure
Okay, let's talk carbonate ion. You know, CO32-? The molecule that's always invited to the chemistry party but ends up causing a bit of a structural headache.
I'm just going to say it: I think drawing its Lewis structure is way more dramatic than it needs to be. Hear me out!
The Carbonate Ion's Identity Crisis
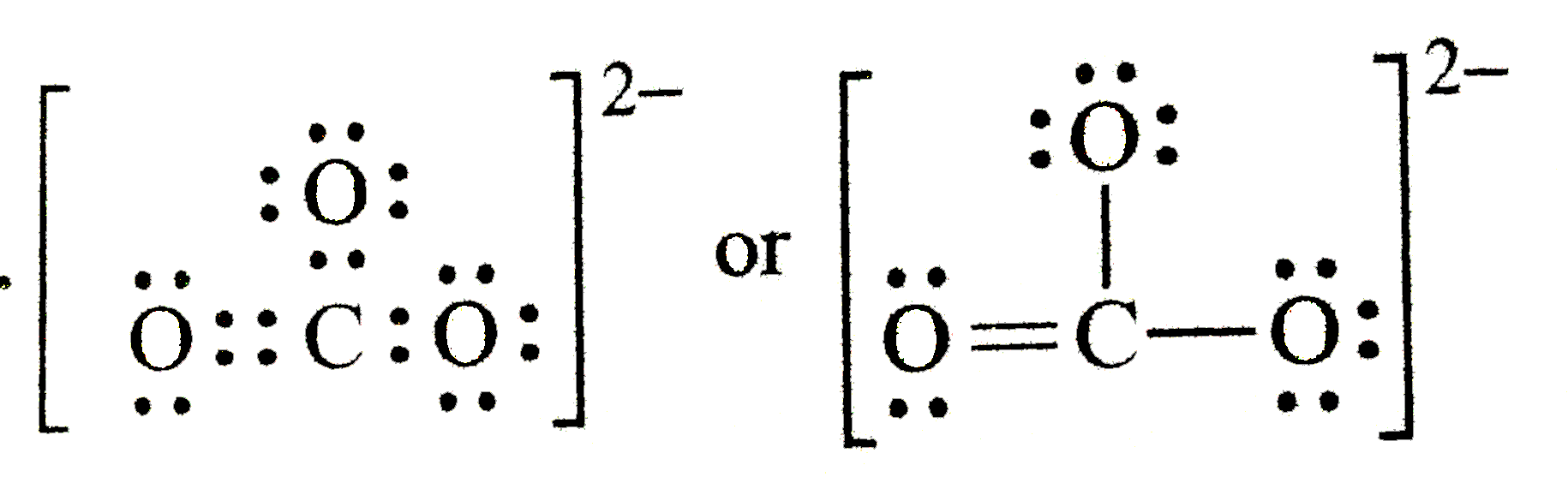
First, we gotta count those valence electrons. Carbon brings four, each oxygen six, and don't forget the two extra from the 2- charge. That's 24 electrons total. Sounds simple enough, right?
Must Read
Carbon in the center, oxygen around it. Easy peasy. Now bond them all with single bonds. But uh oh, someone's still got too many electrons!
The Double Bond Dilemma
This is where it gets spicy. One of those oxygens gets a double bond to carbon. Now everyone thinks it's happy. But is it, really?
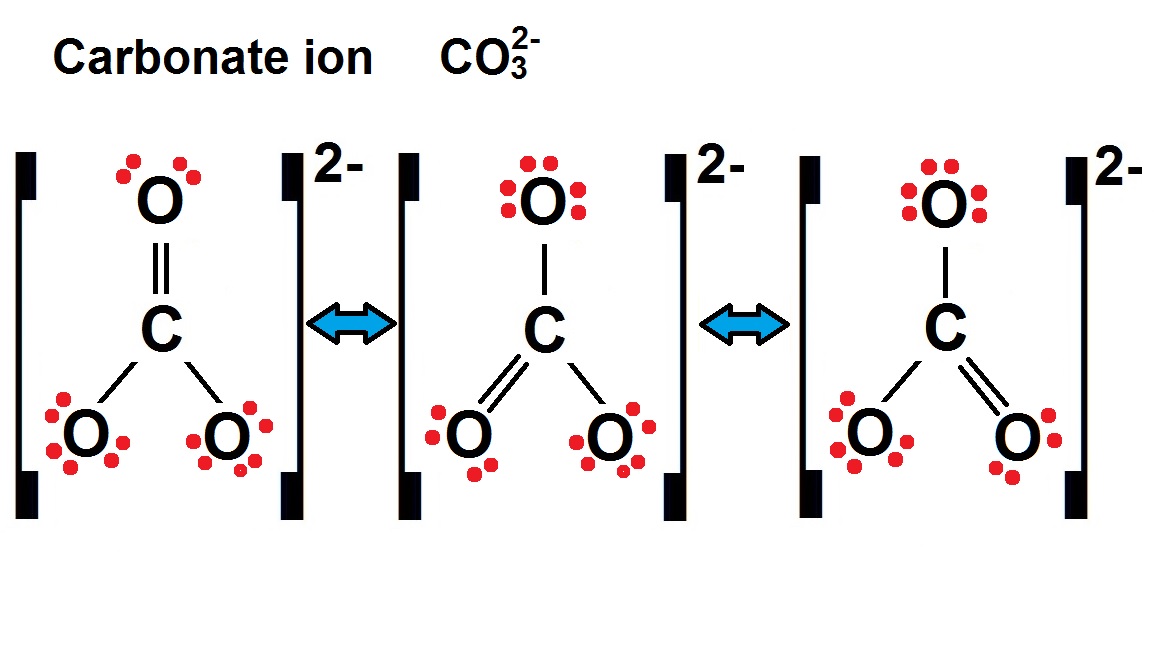
Here's my "unpopular opinion": All three oxygens secretly want that double bond. They're all equally deserving, aren't they? #OxygenEquality

That's where the whole resonance structure thing comes in. You draw it three different ways, double bond rotating around. Showing that the actual structure is some kind of blurry hybrid. A compromise!
Resonance: The "It's Complicated" of Chemistry
Let's be honest, resonance is like a complicated relationship status on Facebook. It's easier to say "it's complicated" than explain the actual nuanced reality.
Three identical structures? Each has single and double bonds switching? Like deciding what to eat for dinner, with endless options and no winning choice!
Chemists insist it's not that the double bond is switching around. They say it's like a blended average. Each oxygen gets a partial double bond. A bit like sharing a pizza... nobody gets a whole slice.
Formal Charges: The Unnecessary Drama
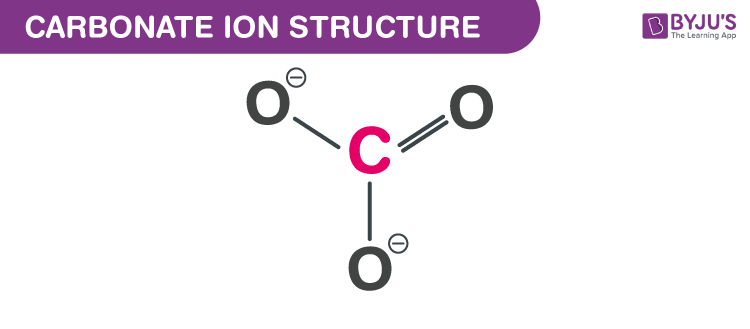
And then come the formal charges. Oxygen with single bond gets a -1, double bonded oxygen gets zero. Carbon gets a +2 (temporarily), but gets two electrons from each oxygen to become zero.
Formal charges are like those awkward family gatherings where everyone's trying to be polite but you can feel the tension in the air. You know it's there.
Do we really need to know how everyone feels in the molecule? Can't we just let them bond in peace?

My Carbonate Ion Conspiracy Theory
Okay, here's where it gets really controversial. I think the carbonate ion is intentionally designed to be confusing. Think about it.
It shows up in everything. Geology (rocks!), biology (bones!), and even everyday life (baking soda!). It's plotting to confuse us all! Well, maybe that is too far.
Maybe it’s just a simple molecule acting as a complicated part of our world! I can accept that.
The Truth Is Out There (Probably)
So, next time you're drawing the Lewis structure for carbonate ion, remember this: you're not alone in your mild frustration.
Embrace the resonance. Accept the formal charges. And maybe, just maybe, whisper a little apology to the oxygens who didn't get the "prime" double-bonded spot.
After all, even molecules deserve a little empathy. (Even if they are plotting world domination, one Lewis structure at a time.)
Just kidding! (Mostly.) But seriously, it's okay to find the carbonate ion a bit dramatic. It's part of its charm. Sort of.
Just don't tell my chemistry professor I said that. Please.
