Carbon Tetrachloride Molar Mass

Alright, buckle up buttercups, because we're diving headfirst into the wild and wacky world of... Carbon Tetrachloride Molar Mass! I know, I know, it sounds like something you'd only hear whispered in the hallowed halls of a chemistry lab (probably while wearing safety goggles that are slightly too big). But trust me, it's way more interesting than watching paint dry... maybe. Okay, probably equally interesting to watching paint dry for some, but I'm gonna try my darnedest to make it feel like watching fireworks!
First things first, what is this mysterious "molar mass" thingamajig? Imagine you're baking a cake. You need the right amount of flour, sugar, and unicorn sprinkles (obviously). Molar mass is kind of like the recipe for a single molecule. It tells you how much that one little molecule "weighs" in the grand scheme of atomic weights. Think of it as the molecule's personal weight on a very, very tiny atomic scale.
Now, let's zoom in on our star of the show: Carbon Tetrachloride! Sounds a bit villainous, doesn't it? Like a rogue agent in a spy movie. (Spoiler alert: it was once used in fire extinguishers and as a cleaning solvent, but those days are long gone due to its... less-than-ideal health effects. Let's just say it's not the kind of spy you want hanging around for cocktails.)
Must Read
The Formula for Fun (and Carbon Tetrachloride!)
Carbon Tetrachloride's chemical formula is CCl4. This tells us exactly what ingredients are in this molecular cocktail: one carbon atom (C) and four chlorine atoms (Cl). It's like a molecular dance party with one carbon doing the tango and four chlorines doing the cha-cha-cha all around it.
To figure out the molar mass, we need to grab our trusty periodic table (or just Google it, no judgment here!). The periodic table is like a cheat sheet that tells you the average atomic mass of each element. Carbon clocks in at about 12.01 atomic mass units (amu), and chlorine weighs in at approximately 35.45 amu.

Let's Crunch Some Numbers (Don't Worry, It's Painless!)
Now for the math! We need to add up the weights of all the atoms in our Carbon Tetrachloride molecule:
(1 x Carbon's weight) + (4 x Chlorine's weight) = Carbon Tetrachloride's Molar Mass
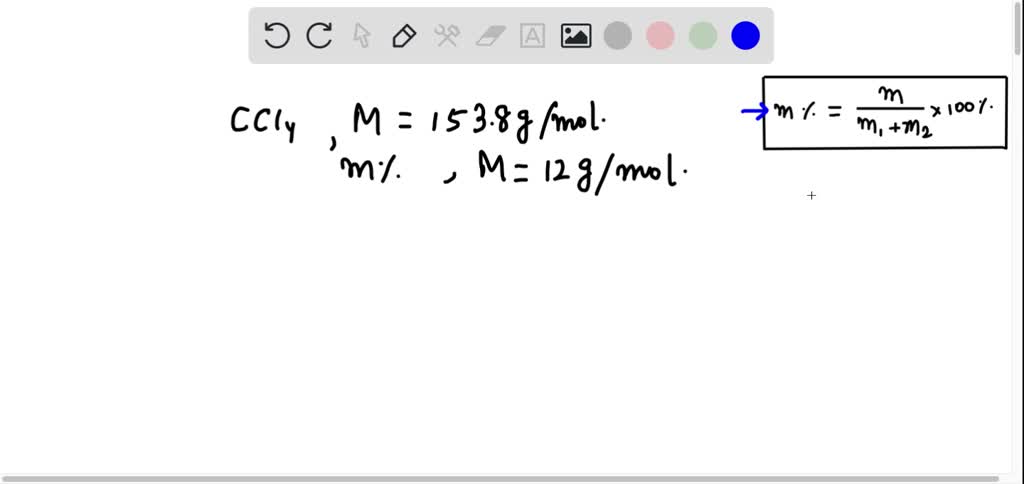
SOLVED: Calculate the mass percent of carbon (C) in carbon
Plugging in those numbers:
(1 x 12.01 amu) + (4 x 35.45 amu) = ???
Get ready to be amazed... (drumroll, please!)... The molar mass of Carbon Tetrachloride is approximately 153.81 amu!

But wait, there's more! Molar mass isn't usually expressed in amu. More commonly, it’s expressed in grams per mole (g/mol). But numerically it's the same.
So, the molar mass of Carbon Tetrachloride is also 153.81 g/mol. This means that one mole (which is a ridiculously large number of molecules, about 6.022 x 1023 – also known as Avogadro's number – but let's not get bogged down in the details!) of Carbon Tetrachloride would weigh approximately 153.81 grams.

Why Should You Care? (Or, Why This Isn't Just Random Chemistry Jargon)
Okay, so maybe you're not planning on brewing up a batch of Carbon Tetrachloride anytime soon (and please, don't!), but understanding molar mass is crucial for anyone working with chemicals. It's the key to calculating how much of a substance you need for a reaction, predicting the yield of a product, and generally making sure you don't accidentally blow anything up (which is always a good goal in chemistry!).
It's like knowing the conversion rate between Euros and Dollars if you are traveling - you need to know what's what when you are working in a different "currency". Molar mass is the "currency exchange rate" between the number of molecules and their mass.
So, the next time you hear someone talking about molar mass, don't run for the hills! Remember our Carbon Tetrachloride adventure, and think of it as just a friendly guide to understanding the weight of the molecular world. And hey, maybe you can even impress your friends at the next party with your newfound knowledge. Just don't try to make them drink Carbon Tetrachloride cocktails. Seriously, don't.