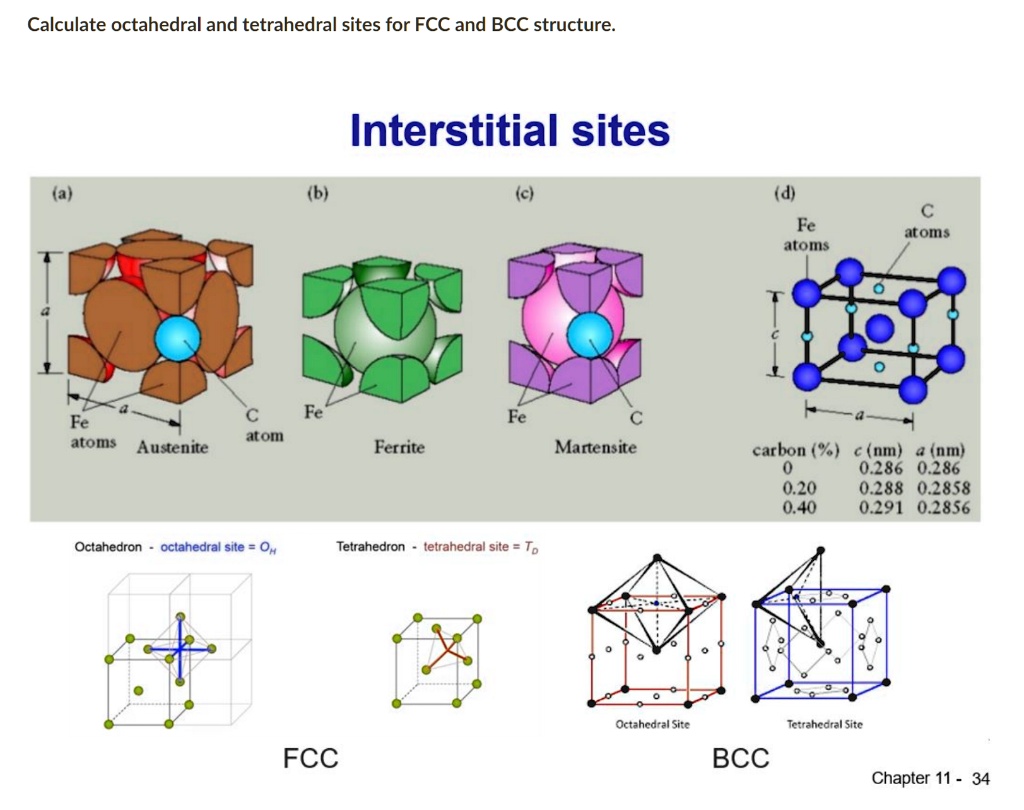
Carbon Migration Barrier Fcc Vs Bcc

Okay, maybe "Carbon Migration Barrier: FCC vs BCC" isn't exactly trending on TikTok, but bear with me! If you've ever wondered about the hidden world of atoms and how they dictate the behavior of materials, specifically how carbon atoms wiggle their way through different crystal structures, then you've stumbled upon something surprisingly fascinating. Think of it as atomic-level choreography, and understanding it can unlock a whole new appreciation for the world around us.
So, why should you, an artist, hobbyist, or simply someone curious about the universe, care about this seemingly complex topic? Well, imagine being able to intuitively understand why some metals are strong and others are brittle, why some rust easily and others don't. This knowledge, gleaned from understanding atomic structures and diffusion, can subtly influence your creative process. Are you a sculptor? Understanding material properties becomes second nature. A jewelry maker? You’ll gain a deeper respect for the metals you manipulate. Even a casual learner will find joy in demystifying the "magic" of everyday materials.
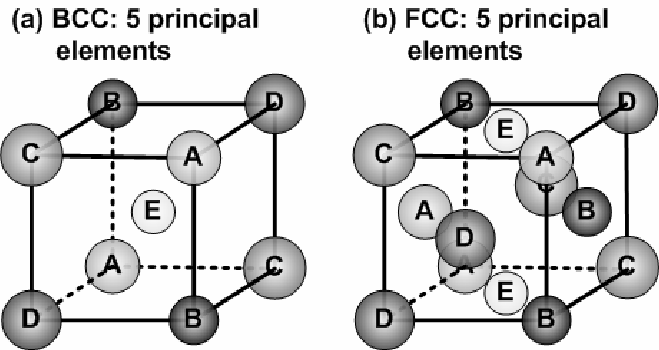
Let's break it down. We're talking about two common crystal structures in metals: FCC (Face-Centered Cubic) and BCC (Body-Centered Cubic). Imagine a bunch of marbles perfectly packed together. FCC has an extra marble in the center of each face of the cube, while BCC has just one in the very center of the cube. Now, picture tiny carbon atoms trying to squeeze their way through these marble arrangements. The "carbon migration barrier" refers to the energy it takes for a carbon atom to jump from one interstitial site (a space between the marbles) to another. In FCC, there are more pathways, albeit potentially more tortuous, compared to BCC. BCC offers a more direct, though potentially more energetically demanding, path. Think of it like navigating a crowded city (FCC) versus forging a path through a dense forest (BCC).
Must Read
The implications are wide-ranging. Think about different types of steel. The arrangement of iron atoms (FCC or BCC) and the presence of carbon directly affect the steel's strength, ductility, and weldability. This impacts everything from the design of bridges and skyscrapers to the creation of intricate metal art. Consider Damascus steel, famed for its beautiful patterns and exceptional strength. The properties are, in part, due to the careful control of carbon diffusion within the iron matrix.
Okay, you're probably thinking, "How can I 'try this at home'?" You're not going to be peering into atomic lattices anytime soon! But you can explore this concept through analogies and simulations. Look for online animations that illustrate FCC and BCC structures. Experiment with building models using marbles or LEGO bricks. Research different types of steel and their applications, paying attention to how their properties relate to their composition. You can even explore materials science kits designed for younger learners, which often simplify these concepts in engaging ways.
Ultimately, understanding the carbon migration barrier in FCC and BCC structures is about more than just memorizing facts. It's about developing a deeper appreciation for the elegant complexity of the material world. It’s about realizing that even seemingly simple objects are governed by intricate atomic interactions. And that realization, my friends, is incredibly satisfying and sparks a continuous journey of learning and discovery. So, dive in, explore, and enjoy the atomic-level adventure!