C2 Paramagnetic Or Diamagnetic

Hey there, science enthusiast! Ever looked at a molecule and wondered if it's secretly magnetic? Today, we're diving headfirst into the fascinating world of molecular magnetism, specifically tackling the burning question: Is C2 (that's carbon diatomic, folks!) paramagnetic or diamagnetic? Get ready for a little molecular dance-off!
First things first, let's decode those fancy terms. Paramagnetic substances are like that friend who's easily influenced – they're weakly attracted to magnetic fields. This happens because they have unpaired electrons buzzing around. Think of them as tiny little magnets themselves, ready to align with an external force. On the flip side, diamagnetic substances are the cool cats who couldn't care less about a magnetic field. They actually repel it slightly! This is because all their electrons are paired up, forming a sort of electromagnetic shield. No unpaired electrons means no individual magnetic moments, and therefore, a little push away from magnetism.
So, C2... The Star of Our Show
Okay, now let's get to the star of our show, C2. Carbon, as you may recall from your periodic table escapades, has 6 electrons. So, C2 has a grand total of 12 electrons to play with. The big question is: how do these electrons arrange themselves within the molecule? Do they pair up nicely, or are there some rebellious, unpaired electrons causing a paramagnetic ruckus?
Must Read
This is where things get a little tricky (don't worry, we'll keep it simple!). You might initially think that C2 has unpaired electrons. After all, if you follow the "normal" filling order of molecular orbitals (sigma before pi!), you'd end up with two electrons occupying the pi bonding orbitals individually before pairing up. And BAM! Unpaired electrons! Paramagnetic C2 confirmed… right? Hold your horses!
Well… not quite. C2 is a bit of a special snowflake. It turns out that for C2, the energy levels of the sigma and pi bonding orbitals are unusually close. Experimental evidence and more sophisticated calculations show that the two pi bonding orbitals fill up before the sigma bonding orbital of slightly higher energy.
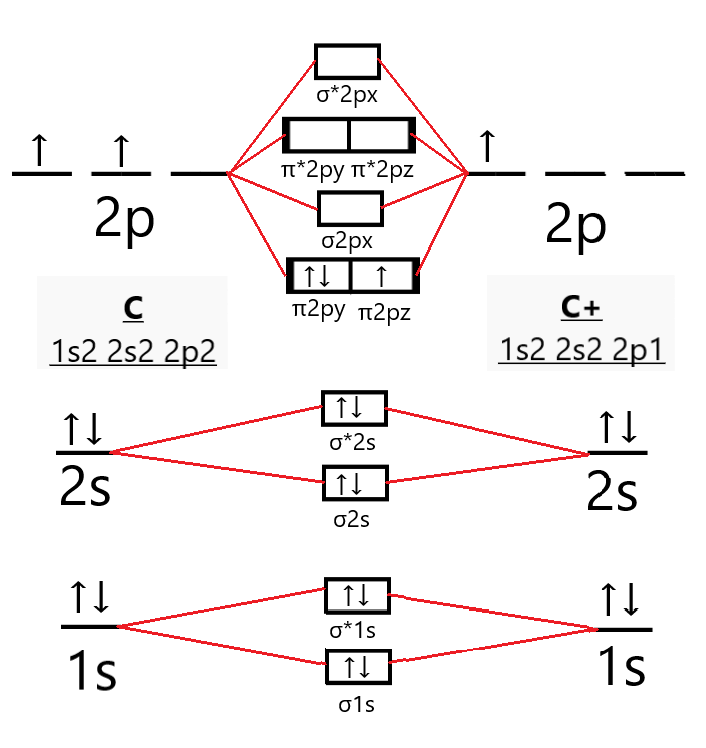
Let me say that again, because it's important. The two pi orbitals fill before that sigma orbital. Why? Well, quantum mechanics is a weird and wonderful place! Let's just say it's due to some complex interactions between the electrons. Scientists use fancy terms like "mixing" and "correlation" to describe these effects, but for our purposes, let's just agree that C2 likes to break the rules.
The Verdict: Diamagnetic Delight!
So, what does this unusual filling order mean? It means that all 12 electrons in C2 happily pair up in the bonding orbitals. No unpaired electrons, no paramagnetic behavior. That’s right: C2 is diamagnetic!
Surprised? Many people are! It’s a great example of how molecular orbital theory can sometimes throw curveballs. And it reminds us that the world of chemistry is full of fascinating surprises. Think about it: a simple molecule like C2, and yet its electronic structure is anything but straightforward!
Bottom line: Don't assume anything when it comes to molecular magnetism. Always double-check those molecular orbital diagrams! And remember, even if a molecule seems like it should be paramagnetic, it might just have a secret diamagnetic identity. It's like finding out your quiet neighbor is actually a professional salsa dancer – unexpected and totally awesome!
So, there you have it! C2, the diamagnetic carbon molecule, defying expectations and keeping things interesting in the world of chemistry. Hopefully, this little exploration has sparked your curiosity and left you with a smile. Now go forth and spread the knowledge… and maybe even impress your friends at your next chemistry-themed party!


