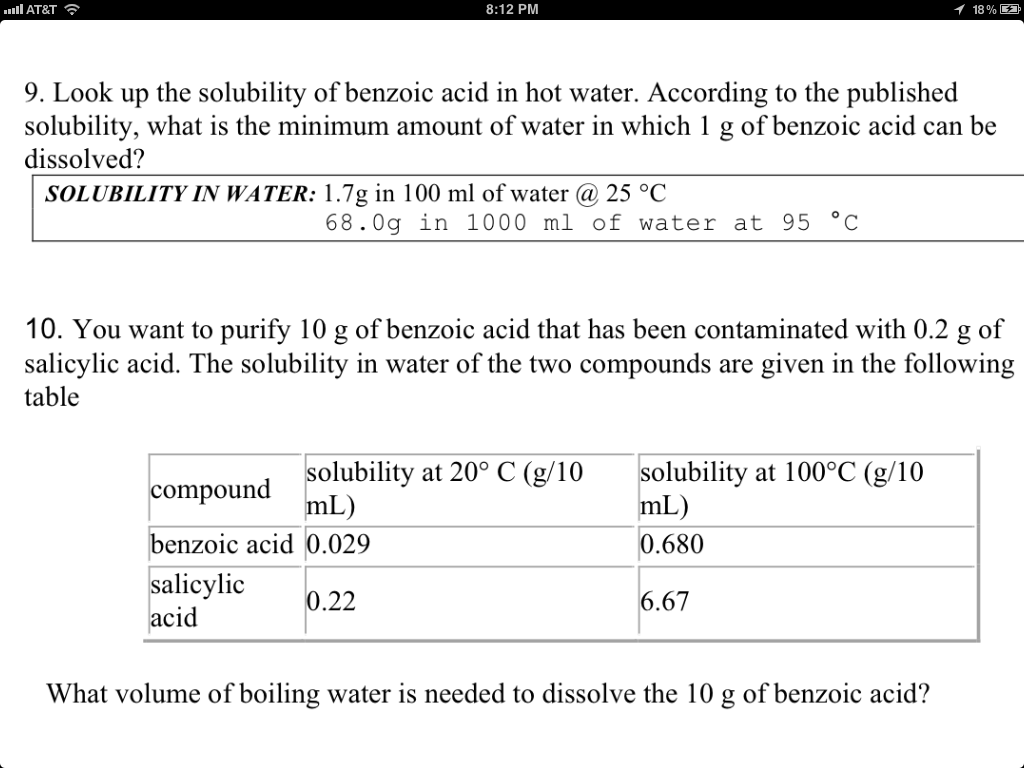
Benzoic Acid Solubility In Water

Hey there, curious minds! Ever wonder what happens when you toss something like benzoic acid into water? Sounds like a high school chemistry class flashback, right? But trust me, this is way more interesting than titrations and beakers. Let's dive into the wild world of benzoic acid solubility!
So, what is benzoic acid anyway? Think of it as a super-powered preservative. It’s a white, crystalline solid, kinda like fancy, edible glitter. Okay, maybe don't eat it straight, but it's used to keep your pickles perky and your sodas… well, soda-y. It keeps the nasties (bacteria, mold) from throwing a party in your food.
Water & Benzoic Acid: A Love-Hate Relationship
Now, for the juicy part: its relationship with water. It’s complicated, to say the least. Imagine trying to mix oil and water. Similar vibe. Benzoic acid is a bit hydrophobic – meaning it's not exactly water's biggest fan. It has a ring-shaped structure (a benzene ring, to be precise) that’s all about keeping to itself.
Must Read
But here's the twist! A tiny bit does dissolve. It’s like that one wallflower at the party who eventually gets dragged onto the dance floor. At room temperature (around 25°C), only about 0.34 grams of benzoic acid will dissolve in a liter of water. That's not a whole lot. Think of it as barely making a splash.
Why such low solubility? Blame it on polarity! Water is a polar molecule – it has a slightly positive end and a slightly negative end. Benzoic acid, on the other hand, is mostly non-polar. Polar likes polar, and non-polar likes non-polar. It’s like trying to mix two different social groups at a school dance.
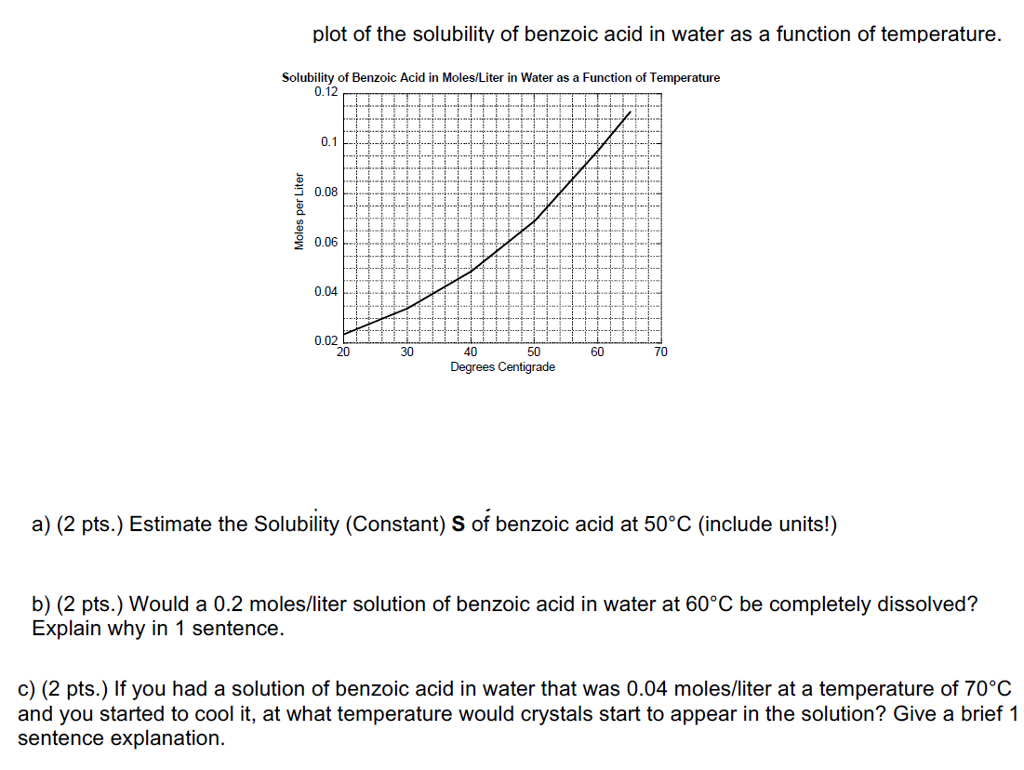
Heat it Up!
Okay, so benzoic acid isn’t thrilled about room temperature water. But what if we crank up the heat? Now things get interesting! The warmer the water, the more benzoic acid will dissolve. Think of it as thawing the ice between them. Heat provides the energy to break the intermolecular forces that are keeping the benzoic acid all bundled up.
This is where the fun begins! It's like convincing the shy benzoic acid to finally come out of its shell. With enough heat, you can dissolve a respectable amount. Not a huge amount, mind you, but a noticeable improvement. It’s like going from barely being able to hear the music to tapping your foot along to the beat.
The pH Factor: Acidity Rules!
Another quirky fact: pH plays a role! Benzoic acid is, well, an acid. That means it can donate a proton (H+). When it does that, it becomes benzoate, an ion with a negative charge. Charged particles love water. So, increasing the pH (making the solution more alkaline) can dramatically increase benzoic acid's apparent solubility. It's a sneaky chemical trick!

Think of it as putting on a disguise. Benzoic acid is shy and doesn't want to dance. But benzoate? Benzoate is ready to boogie! Changing the pH is like giving benzoic acid a confidence boost and a brand new outfit.
Why Should You Care?
Alright, so benzoic acid's dissolving habits might not seem like the most thrilling topic. But understanding solubility is crucial in all sorts of fields! From pharmaceuticals (getting drugs to dissolve in the body) to food science (preserving your favorite snacks), it's everywhere.

Plus, it’s just cool to know how things work at a molecular level. It's like peeking behind the curtain to see the wizard pulling the levers. Next time you’re sipping a fizzy drink or enjoying a perfectly preserved jar of pickles, you can thank (or blame!) the principles of solubility and the complicated relationship between benzoic acid and water. You'll also be able to drop a bomb of knowledge at your next trivia night!
So, there you have it. Benzoic acid solubility: not as boring as it sounds! It's a story of shyness, heat-induced breakthroughs, and pH-powered makeovers. Keep exploring, keep questioning, and keep having fun with chemistry!
One last thing, remember that it's always a good idea to double-check safety information when dealing with chemicals and experiments. Stay safe and stay curious!