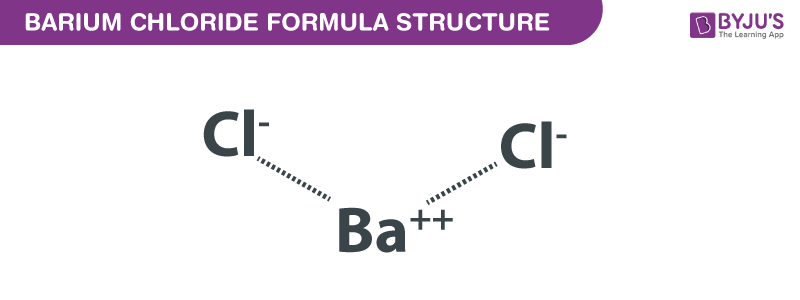
Barium Chloride Molecular Mass

Ever heard of Barium Chloride? Sounds intimidating, right? Like something out of a sci-fi movie. But trust me, it's more interesting than scary! One of the key things about it is its molecular mass. Think of it as the compound's own personal weight. So, why are we talking about this weight? Because it unlocks all sorts of cool secrets!
Decoding the Weight: The Molecular Mass Mystery
Okay, let's break it down. The molecular mass of Barium Chloride is roughly 208.23 atomic mass units (amu). Don't panic! That's just a fancy way of saying how heavy a single molecule of this stuff is. Imagine each atom is a building block. Barium is one type of block, and Chlorine is another. You need to know how much each block weighs to find the overall weight of the whole molecule.
Why should you care? Well, understanding this mass is like having a secret decoder ring for chemical reactions. It tells us how much of Barium Chloride we need to react with other substances. It’s fundamental for chemistry experiments. Picture it as following a cake recipe, you need the exact amount for each ingredient to make the perfect cake.
Must Read
The Atomic Weight Workout
Where does this number come from? The periodic table, of course! That's the cheat sheet for all things atomic. Barium (Ba) weighs about 137.33 amu. Chlorine (Cl) tips the scales at roughly 35.45 amu. But wait! Barium Chloride is BaCl2. That little "2" means we have two chlorine atoms. So, we need to double chlorine's weight. Two times 35.45 equals 70.90 amu.
Now, just add them together: 137.33 + 70.90 = 208.23 amu. Boom! We've calculated the molecular mass. Isn't that like a chemical math magic trick?
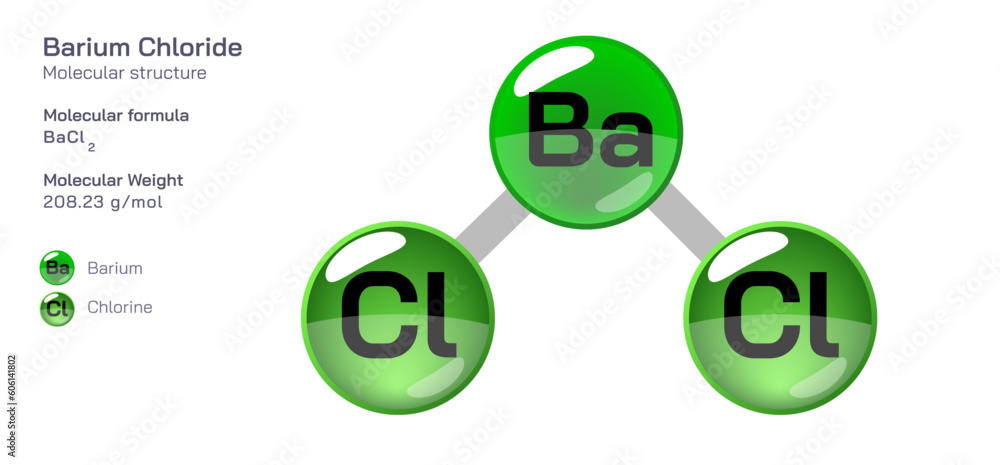
Think of it like building a Lego structure. You know the weight of each Lego brick, so you can easily calculate the weight of the whole model. Barium Chloride's molecular mass is the same idea, but with atoms!
Beyond the Classroom: Real-World Fun
So, it's not just about boring chemistry textbooks. Knowing the molecular mass of Barium Chloride is vital in many real-world applications. Let’s say you're testing for sulfate ions in water. Barium Chloride reacts with sulfates to form a precipitate. Without knowing the molecular mass, how would you calculate the amount of Barium Chloride needed to perform the test correctly?
Or, imagine you're a scientist developing new materials. You might be using Barium Chloride as a precursor. Accurately determining the amount needed requires knowledge of its molecular weight. It’s the backbone of research and experiments!

Why Barium Chloride is a Rockstar
What makes Barium Chloride so special? It's all about its unique properties. It's a salt that dissolves in water, producing barium ions and chloride ions. These ions are pretty reactive, leading to many exciting chemical reactions.
But remember, safety first! Barium Chloride can be toxic, so it's essential to handle it with care in a lab setting. Think of it as a powerful tool that needs respect and caution. Treat it like a delicate instrument, and you'll be fine.

Barium Chloride might sound complex, but it's really just a molecule with a specific weight, a molecular mass. This mass is a key that unlocks so many scientific possibilities. From lab experiments to real-world applications, it’s a crucial piece of the chemical puzzle.
Dive Deeper!
So, are you ready to explore more about Barium Chloride? There's a whole world of fascinating chemistry waiting for you. Don't be afraid to dive in and discover something new. Who knows, you might just find your inner scientist!
"The important thing is to never stop questioning." - Albert Einstein
Think of the molecular mass of Barium Chloride, or any chemical compound, as a gateway. It’s a gateway to understanding the universe at a molecular level. Go forth and explore!