Barium Acetate Molar Mass
Ever stumbled upon something so seemingly dull it practically begged you to ignore it? Well, get ready, because we're diving into the captivating world of Barium Acetate and its molar mass! Trust me, it's not as snoozeworthy as it sounds.
Barium Acetate: A Name That Demands Respect (Sort Of)
First, let's meet our star: Barium Acetate. It sounds like a character from a Victorian novel, doesn’t it? Maybe a stern but secretly kind professor who unravels mysteries in his spare time.
But alas, it's a chemical compound, a white crystalline solid. Don't let the "solid" part fool you; it's far from boring.
Must Read
Molar Mass: The Weight of the World (Well, Barium Acetate's World)
Now, molar mass. This is where things get…interesting. It's essentially the weight of one mole (a specific number) of a substance. Think of it like the average weight of a single student in a very, very large class.
Imagine trying to weigh each student individually! It's easier to find the average weight to understand the overall weight contribution.
Why Bother With Molar Mass Anyway?
Why do we care about the molar mass of Barium Acetate? Because it’s fundamental! It’s like knowing the cost of a single widget when you’re buying a whole factory full of them.
Need to create a specific amount of a solution for an experiment? Molar mass is your guide. It’s the key to unlocking precise chemical reactions and avoiding disastrous explosions (hopefully!).

The Great Molar Mass Calculation Adventure
So, how do we actually find the molar mass? Prepare for a thrilling mathematical quest! (Okay, maybe not thrilling, but definitely important.)
You’ll need a periodic table. That trusty chart of elements you probably last saw in high school. It's time to dust it off and renew acquaintances.
Each element has an atomic weight listed. Find Barium (Ba), Carbon (C), Hydrogen (H), and Oxygen (O). These are the players in our Barium Acetate drama.
Barium Acetate has the chemical formula Ba(C2H3O2)2. That little subscript "2" means that the acetate group (C2H3O2) is present twice.

Let's break it down. One Barium atom, four Carbon atoms, six Hydrogen atoms, and four Oxygen atoms. The math is coming… brace yourselves!
Multiply the number of each atom by its atomic weight from the periodic table:
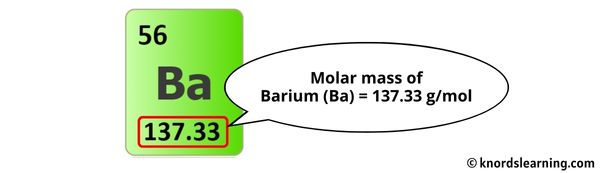
- Barium (Ba): 1 x 137.33 g/mol = 137.33 g/mol
- Carbon (C): 4 x 12.01 g/mol = 48.04 g/mol
- Hydrogen (H): 6 x 1.01 g/mol = 6.06 g/mol
- Oxygen (O): 4 x 16.00 g/mol = 64.00 g/mol
Now, add those all up! 137.33 + 48.04 + 6.06 + 64.00 = 255.43 g/mol.
Voila! The molar mass of Barium Acetate is approximately 255.43 g/mol. You've done it! Celebrate with a non-chemical-related treat.
The Surprising Applications of Barium Acetate (and its Molar Mass!)
Where does Barium Acetate pop up in the real world? You might be surprised. While it's not exactly a household name, it plays a role in various industries.
It can be used in dyeing textiles, manufacturing other chemicals, and even in certain types of ceramics. The molar mass is crucial for ensuring the right proportions in these processes.
Imagine a textile dyer meticulously calculating the amount of Barium Acetate needed to achieve the perfect shade of blue. All thanks to understanding molar mass!
Barium Acetate: More Than Just a Number
Okay, so Barium Acetate and its molar mass might not be the most glamorous topic. But hopefully, you see that even seemingly dry scientific concepts can have a surprising story to tell.

It underscores the importance of precision and understanding in the world around us. Every tiny detail, every little calculation, contributes to something bigger.
Next time you encounter a chemical formula, remember the hidden adventures within. Remember the molar mass calculation and the textile dyer striving for the perfect shade of blue.
The Final Word on Molar Mass
Molar mass is the unsung hero of chemistry. It may not win any popularity contests, but it’s essential for countless applications that impact our daily lives.
So, embrace the numbers, explore the periodic table, and never underestimate the power of a good calculation. You never know what exciting discoveries await you in the world of chemistry!
Remember that it all starts with understanding the fundamentals, even the seemingly simple concept of molar mass. It all adds up.
