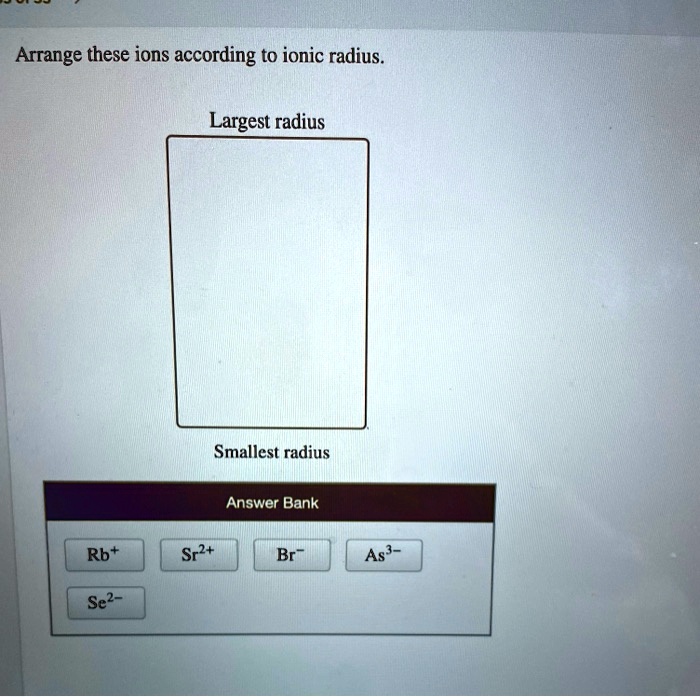
Arrange These Ions According To Ionic Radius.

Okay, chemistry buffs and curious minds, let’s dive into something that might sound intimidating, but is actually kinda cool: ionic radius. We’re talking about arranging ions by size. Think of it as Marie Kondo-ing your elements, but instead of sparking joy, it's all about electron clouds and nuclear pull. Ready to declutter some ions?
First Things First: What's an Ion, Anyway?
Before we get into the nitty-gritty of arranging, let's recap. An ion is simply an atom or molecule that has either gained or lost electrons, giving it a net electrical charge. If it loses electrons, it becomes a positively charged cation (think: cats are positive… kinda!). If it gains electrons, it becomes a negatively charged anion.
Imagine a social butterfly (an atom) surrounded by friends (electrons). If it gives away some friends, it becomes more “positive” because it has more people vying for its attention. If it collects more friends, it becomes more “negative” because it’s suddenly more exclusive. Okay, maybe that’s a bit of a stretch, but you get the idea!
Must Read
The Ionic Radius Lowdown
The ionic radius is essentially the size of an ion. But here’s the catch: ions aren't always the same size as their neutral atom counterparts. Losing electrons (forming a cation) generally makes an ion smaller. Why? Fewer electrons mean less electron-electron repulsion, and the nucleus pulls the remaining electrons in tighter. Think of it like losing weight – you become more compact!
Conversely, gaining electrons (forming an anion) generally makes an ion larger. More electrons mean more electron-electron repulsion, causing the electron cloud to expand. It's like putting on a cozy, oversized sweater – you take up more space.
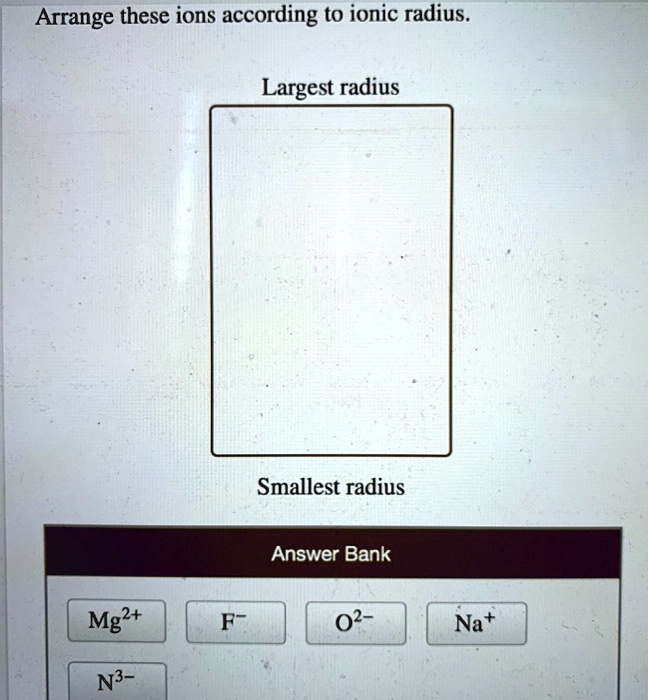
The Arrangement Game: Rules of Engagement
So, how do we actually arrange these ions according to size? Here's a simplified roadmap:
- Look at the Charge: Generally, more negatively charged ions are larger, and more positively charged ions are smaller. So, Cl- will likely be larger than Na+.
- Consider the Number of Electrons: For ions with the same charge (isoelectronic species), the ion with more protons (higher atomic number) will be smaller. More protons mean a stronger positive charge in the nucleus, pulling the electrons in tighter.
- Periodic Trends: Remember your periodic table! Ionic radius generally increases down a group (column) because you're adding more electron shells. It generally decreases across a period (row) because the nuclear charge increases, pulling electrons closer.
Practical Tip: When comparing ions of the same element, the trend is almost always clear-cut. For example, Fe3+ will always be smaller than Fe2+, which will always be smaller than Fe.
Example Time! Let's Get Hands-On
Let's say we need to arrange these ions in order of increasing ionic radius: K+, Cl-, Ca2+, S2-.
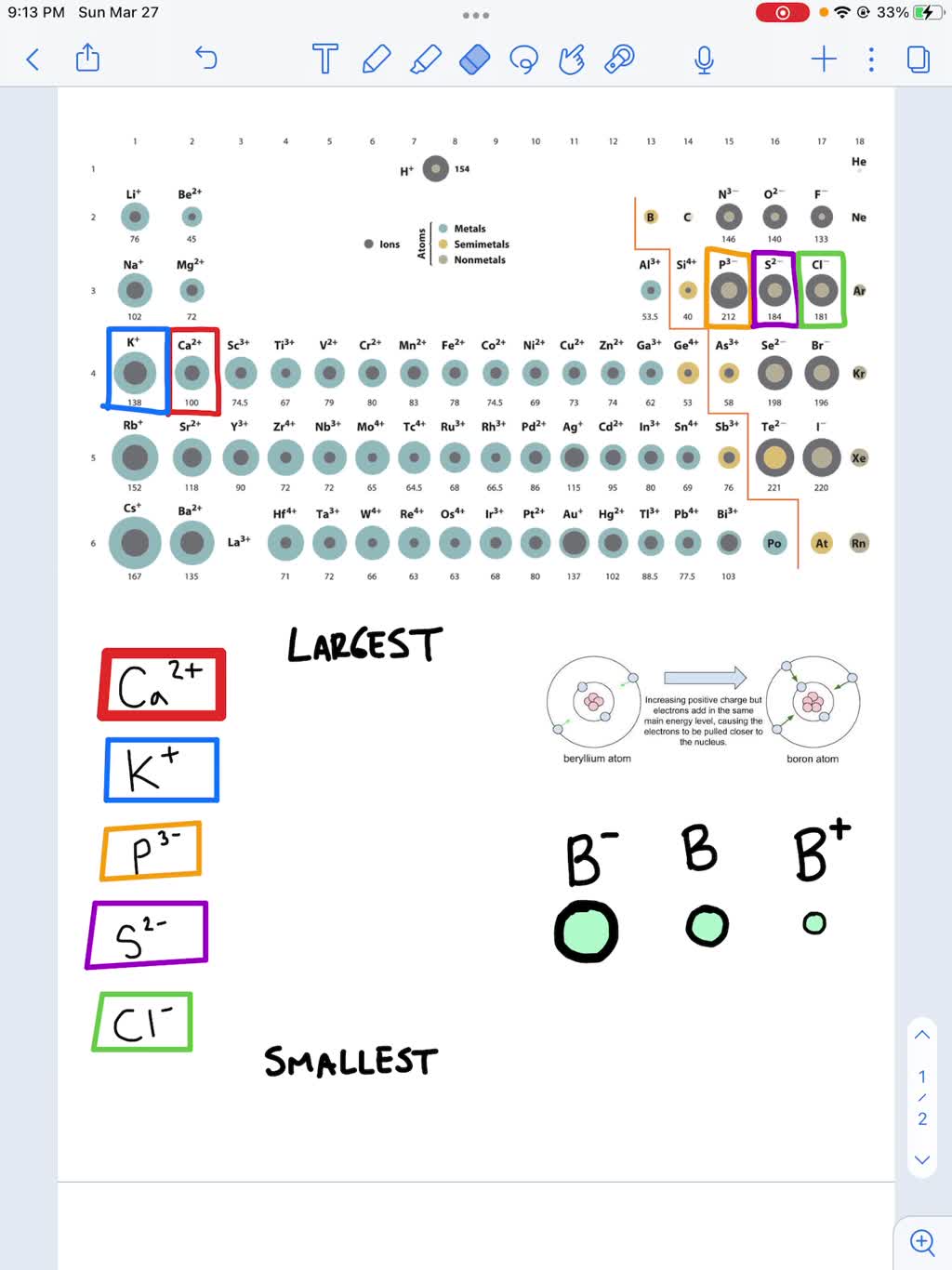
- Charge: S2- and Cl- are anions (negative), so they'll likely be larger than K+ and Ca2+ (cations, positive).
- Number of Electrons & Periodic Trends: These ions are all isoelectronic (they have the same number of electrons). Therefore, we look at the number of protons. Ca has the highest number of protons, then K, then Cl, then S. More protons means a greater pull on the electrons.
Therefore, the order of increasing ionic radius is: Ca2+ < K+ < Cl- < S2-.
Why Bother? The Real-World Connection
You might be thinking, “Okay, cool… but why should I care?” Well, ionic radius plays a critical role in many chemical and biological processes. It affects the strength of ionic bonds in compounds, the conductivity of electrolytes in batteries, and even the selectivity of ion channels in your nerve cells (allowing your brain to send signals!).

Imagine trying to fit a square peg (a large ion) into a round hole (a small binding site). It just won't work! Ionic radius dictates how well ions interact with other molecules, influencing everything from drug efficacy to the formation of minerals.
Fun Fact: The size of ions even influences the color of certain gemstones! The presence of specific ions with varying radii affects how light interacts with the crystal structure, giving rise to vibrant hues.
A Moment of Reflection
Thinking about ionic radius, and how even the smallest differences in size and charge can have profound effects, makes you appreciate the intricate balance that governs the world around us. It’s a reminder that even seemingly insignificant details can be crucial in shaping our reality. Just like carefully arranging your priorities in life can lead to a more fulfilling existence, understanding the arrangement of ions helps us to understand the fundamental building blocks of the universe. And that's a pretty cool thought, isn’t it?