Ammonium Sulfate Molar Mass

Okay, chemistry isn't usually the first thing that springs to mind when you’re scrolling through Instagram, right? But hear me out. Understanding something seemingly obscure like the molar mass of ammonium sulfate can actually be pretty fascinating, and even...useful? Think of it as unlocking a secret level in the game of life. Stay with me!
So, what exactly is ammonium sulfate? Chemically speaking, it's (NH₄)₂SO₄. In plain English, it's a salt containing ammonium and sulfate ions. You'll find it hanging out in a bunch of different places, especially in agriculture. Think of it as a fertilizer superhero, helping plants grow big and strong. It's also used in industrial processes like water treatment and even food production. Who knew, right?
Breaking Down the Numbers: Molar Mass 101
Let’s talk about molar mass. This is essentially the weight of one mole (a specific quantity, like a dozen but for atoms) of a substance. Knowing the molar mass of ammonium sulfate is crucial for all sorts of calculations, like figuring out how much fertilizer to use or how much of it you need for a chemical reaction.
Must Read
The molar mass of ammonium sulfate is approximately 132.14 grams per mole (g/mol). But how do we arrive at that number? Let's break it down:
- Nitrogen (N): The molar mass of nitrogen is roughly 14.01 g/mol. We have two ammonium ions (NH₄), so that's 2 x 1 x 2 = 2 nitrogen atoms.
- Hydrogen (H): The molar mass of hydrogen is around 1.01 g/mol. Each ammonium ion has four hydrogen atoms, and we have two ammonium ions, for a total of 2 x 4 = 8 hydrogen atoms.
- Sulfur (S): The molar mass of sulfur is about 32.07 g/mol. There's only one sulfur atom in ammonium sulfate.
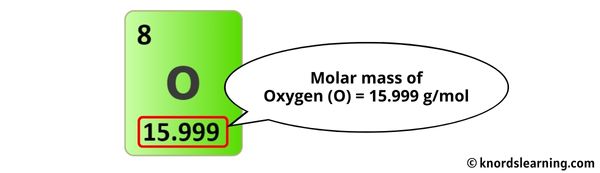
- Oxygen (O): The molar mass of oxygen is approximately 16.00 g/mol. We have four oxygen atoms in the sulfate ion.
Now, we just add it all up:

(2 x 14.01) + (8 x 1.01) + (1 x 32.07) + (4 x 16.00) = 132.14 g/mol
Voila! We've cracked the code.
Why Should You Care? Practical Applications
Okay, so maybe you’re not planning on becoming a fertilizer magnate anytime soon. But understanding molar mass actually has some surprisingly relatable applications. For example:
- Gardening: If you're using ammonium sulfate as a fertilizer (always follow instructions carefully!), knowing its molar mass helps you calculate the correct dosage to nourish your plants without overdoing it. Think of it like following a recipe to bake the perfect cake – too much or too little of an ingredient can ruin the whole thing.
- Cleaning: Ammonium sulfate can be found in some cleaning products. Understanding the concentration (which relies on molar mass calculations) helps you use the product safely and effectively.
- Understanding Food Labels: While ammonium sulfate isn't something you'd find listed as an ingredient (usually), understanding the principles of chemical composition helps you decipher food labels and be a more informed consumer. Knowledge is power, after all!
Fun Fact Interlude
Did you know that ammonium sulfate used to be a byproduct of coal production? It's a testament to how our understanding and use of resources have evolved over time. Now, it's primarily synthesized in factories.

Connecting Chemistry to Everyday Life
We often think of chemistry as something confined to labs and textbooks, but it's actually all around us. From the air we breathe to the food we eat, chemical reactions and compounds are constantly at play. Understanding the basics, like the molar mass of ammonium sulfate, helps us appreciate the invisible science that shapes our world.
So, the next time you're tending to your garden, remember the humble ammonium sulfate and the (not so humble) molar mass that makes it all work. It's a reminder that even seemingly complex scientific concepts can have practical, everyday relevance. And who knows, maybe you'll even impress your friends with your newfound chemical knowledge at your next dinner party!

