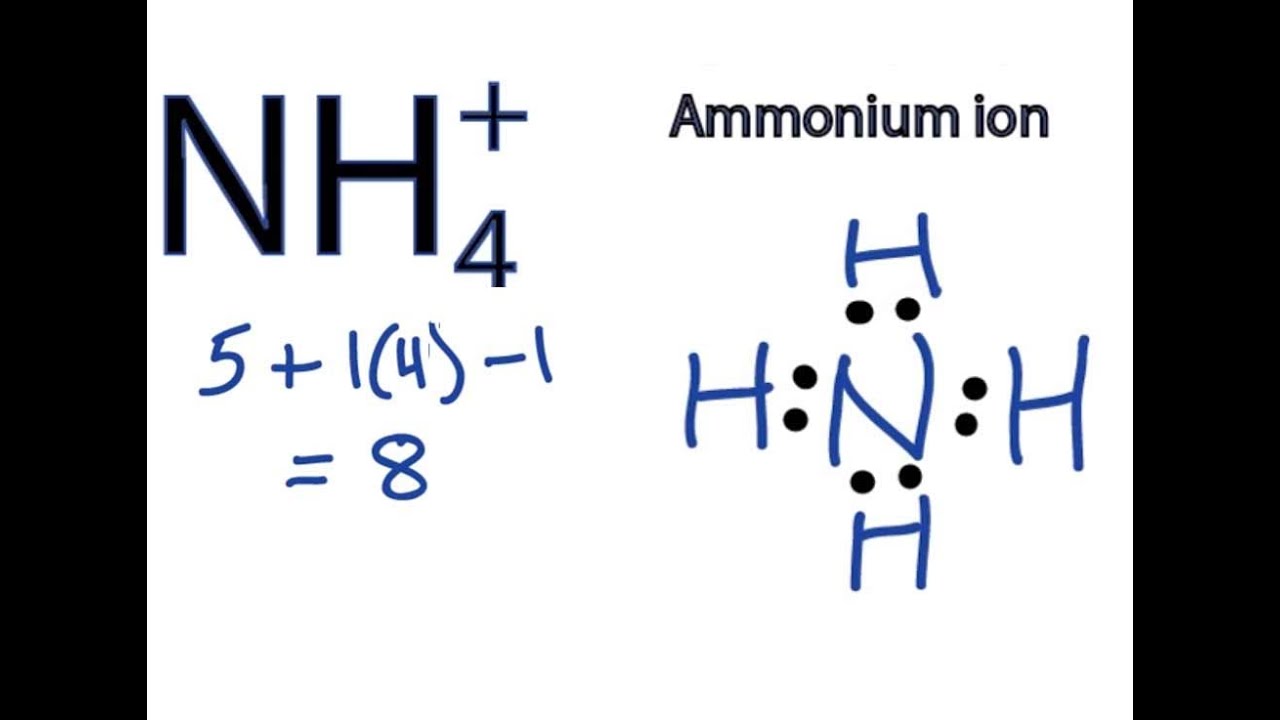
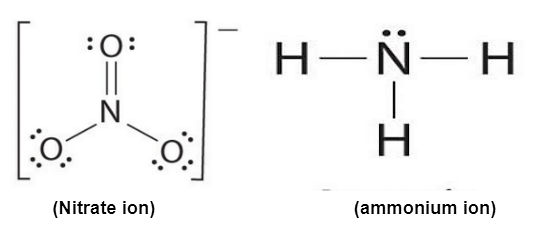Ammonium Ion Lewis Structure
Ever wonder how fertilizers help plants grow or why certain cleaning products work so well? Often, the answer lies in the intricate world of chemical structures, and today we're diving into a particularly interesting one: the ammonium ion (NH₄⁺). Now, drawing chemical structures might sound intimidating, but trust me, understanding the ammonium ion's Lewis structure is like unlocking a secret code to understanding a little bit of chemistry that's all around you.
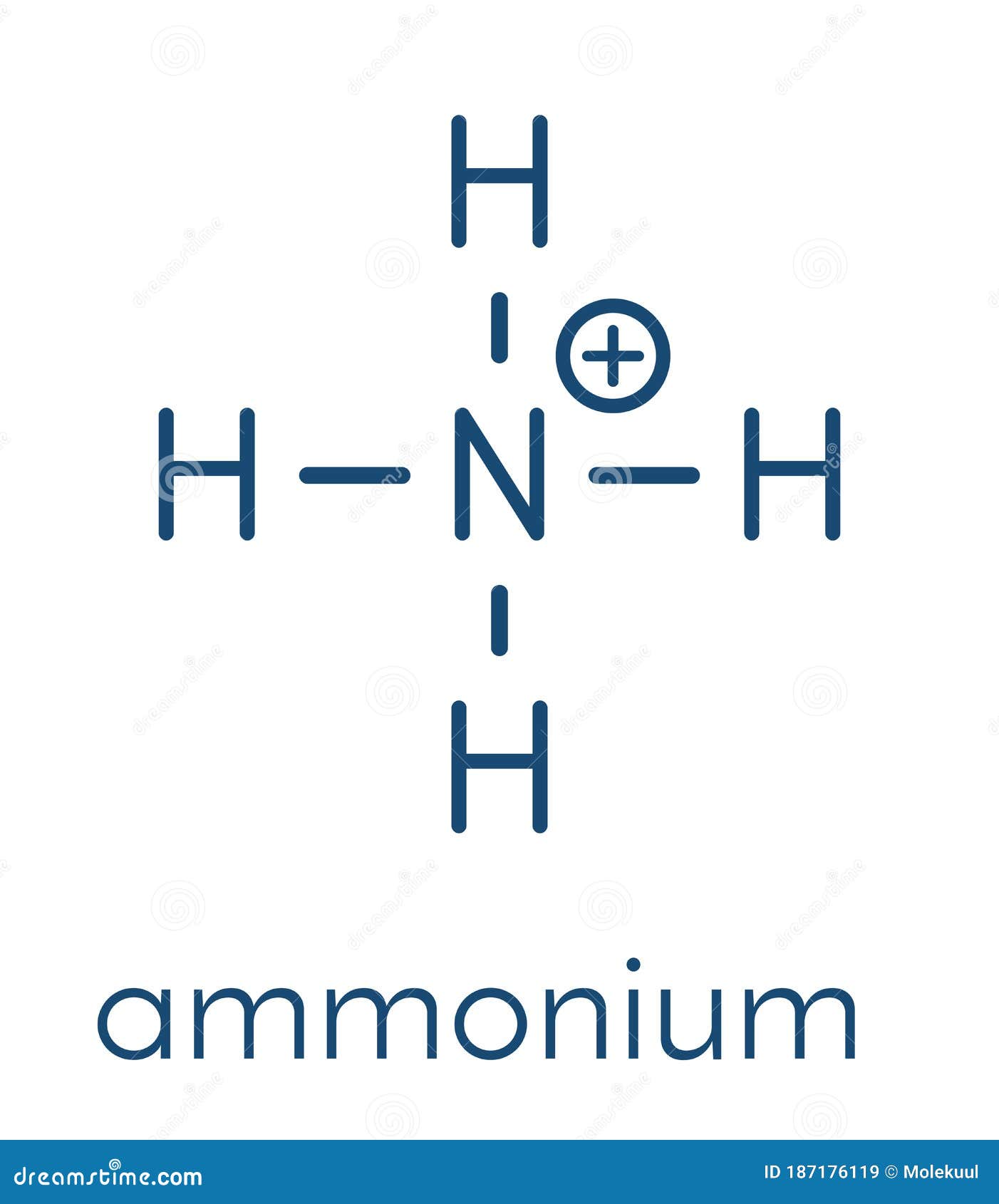
So, what's a Lewis structure, and why bother with it? Think of it as a visual roadmap of a molecule or ion. It shows us how atoms are connected and, crucially, where the electrons are. Remember electrons? They're the tiny particles that dictate how atoms bond together. For ammonium, the Lewis structure helps us visualize how four hydrogen atoms bond to a single nitrogen atom, and why the whole thing carries a positive charge. This positive charge is what makes ammonium interact with other molecules, giving it its unique properties.
The main benefit of understanding the ammonium ion's Lewis structure is that it provides a clear and simple way to represent its bonding. It helps predict the shape of the ion (which is tetrahedral, by the way!), its reactivity, and its interaction with other molecules. This visual representation simplifies a complex concept and makes it easier to grasp.
Must Read
Where can you see ammonium in action? Think about fertilizers used in agriculture. Ammonium compounds, like ammonium nitrate, are essential sources of nitrogen, a crucial nutrient for plant growth. The ammonium ion in these fertilizers provides plants with readily available nitrogen they can absorb and use to build proteins and other essential molecules. In education, drawing the Lewis structure of ammonium is a common exercise in introductory chemistry courses. It reinforces the concepts of covalent bonding, formal charge, and the octet rule (which describes how atoms "want" eight electrons in their outer shell). Even in household cleaning products, you might find ammonium hydroxide, a weak base that helps dissolve grease and grime. Its effectiveness stems from the properties of the ammonium ion it contains.
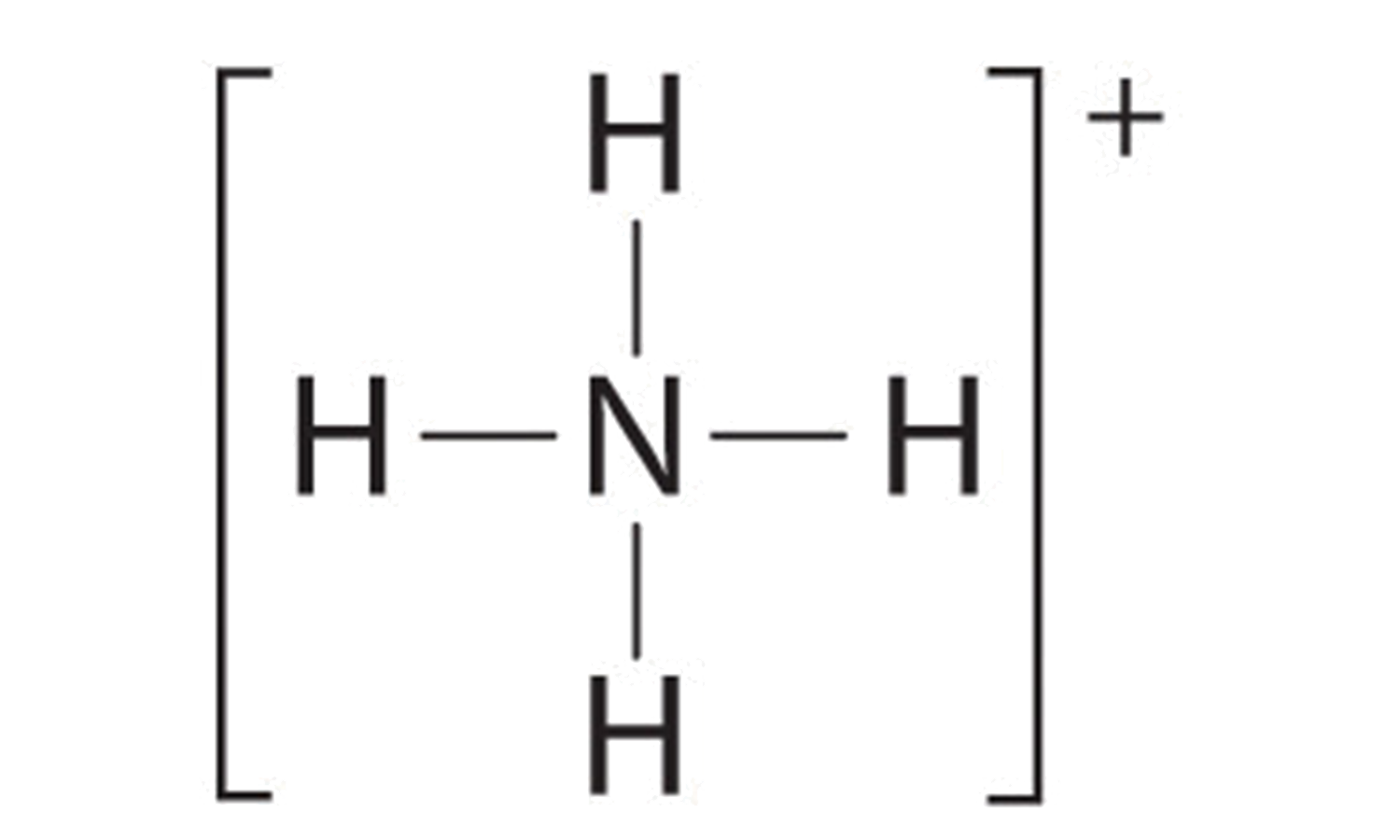
Want to explore the ammonium ion's Lewis structure yourself? Here are a few simple tips: 1) Start by knowing the number of valence electrons (electrons in the outermost shell) for each atom. Nitrogen has 5, and hydrogen has 1. 2) Remember the overall charge of the ammonium ion is +1, meaning it has lost one electron. 3) Draw the nitrogen atom in the center and arrange the four hydrogen atoms around it. 4) Use lines to represent covalent bonds (shared electrons) between nitrogen and each hydrogen. 5) Finally, enclose the entire structure in brackets and write a "+" superscript to indicate the positive charge. You can find plenty of online resources and videos that walk you through the process step-by-step.
So, the next time you see a bag of fertilizer or read about a chemical reaction, remember the humble ammonium ion and its revealing Lewis structure. It's a tiny key that unlocks a world of chemical understanding!