Alkaline Earth Metals In Periodic Table
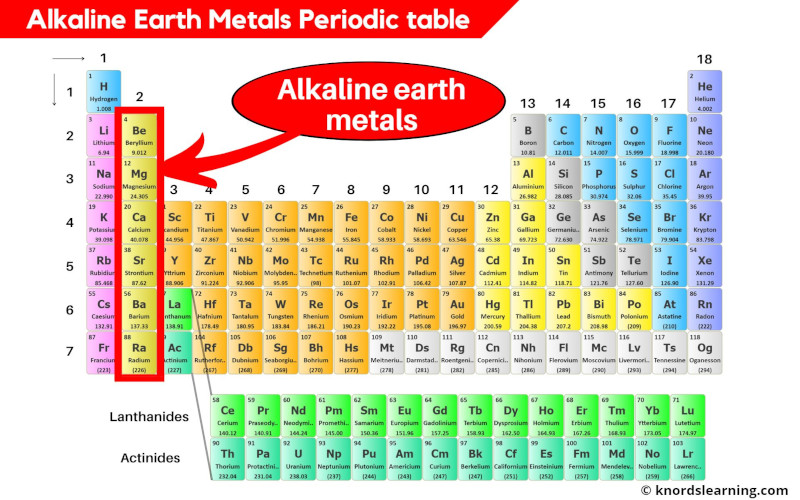
Ever heard of the Alkaline Earth Metals? They're a group of elements hanging out together in the periodic table's neighborhood, specifically in Group 2. Think of them as the slightly less wild cousins of the super-reactive Alkali Metals in Group 1.
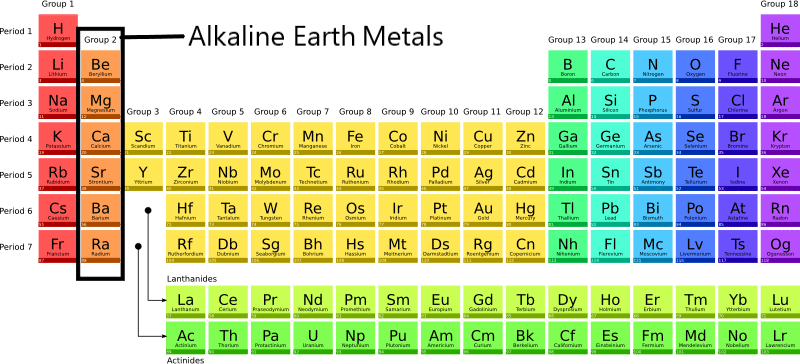
A Family Portrait: Meet the Alkaline Earth Metals
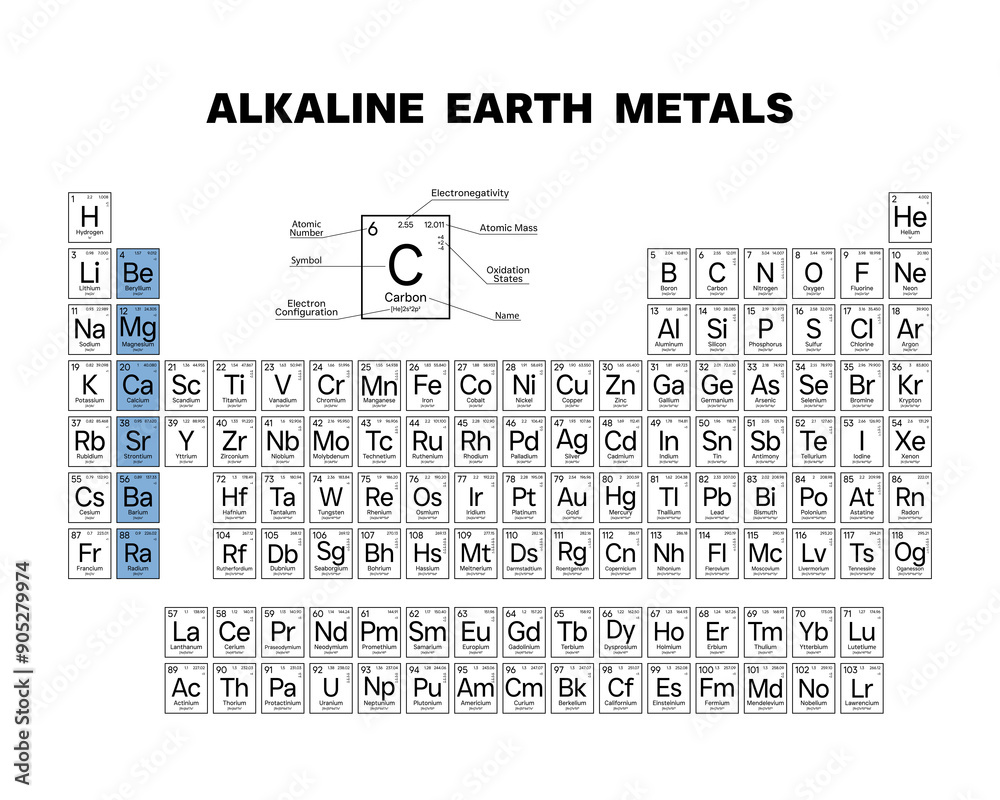
This family includes Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), and Radium (Ra). Radium is radioactive, so you wouldn't want to invite it to a party!
Let's break down what makes them so special and, dare I say, kinda cool. They're all metals, which means they're generally shiny, good conductors of electricity, and love to form positive ions.
Must Read
The Key to Their Personality: Two Electrons
These elements all have two electrons in their outermost shell. This is their defining characteristic.
They are always eager to give these electrons away to form a bond, making them reactive (but not quite as reactive as the Alkali Metals).
Magnesium: The Multitasker of Metals
First up, let's talk about Magnesium! This stuff is everywhere. You probably know it from those fizzy magnesium supplements people take after a workout.
Magnesium is crucial for healthy muscles and nerves. It's also a key ingredient in chlorophyll, which is what makes plants green and able to perform photosynthesis.
And guess what? Magnesium is used in alloys to make them lighter and stronger. Ever held a super light but sturdy laptop? Thank magnesium!
Calcium: Bones, Bones, and More Bones!
Next, we have the mighty Calcium. Everyone knows that calcium is important for strong bones and teeth.
It's like the body's construction worker, constantly rebuilding and reinforcing our skeletal structure. So, drink your milk!
But calcium does way more than just build bones. It also plays a vital role in muscle function, nerve transmission, and blood clotting. Seriously, it's a busy element!
Beryllium: The Lightest of the Bunch
Then there is Beryllium, a lightweight champion. It's known for its high strength-to-weight ratio.
That makes it perfect for aerospace applications. Think satellites and high-speed aircraft!
However, beryllium can be toxic, so you won't find it in many consumer products. Handle with care, folks!
Strontium: The Fireworks Star
Let's not forget about Strontium! This alkaline earth metal loves to put on a show.
Strontium compounds are responsible for the vibrant red color in fireworks. Imagine a Fourth of July without that brilliant red – boring!
Strontium is also used in some toothpastes to help reduce tooth sensitivity. Who knew fireworks could help your teeth?

Barium: The X-Ray Helper
Now for Barium, an element with a heavy presence. Barium sulfate is often used in medical imaging, specifically X-rays.
Patients drink a "barium cocktail" which helps to improve the visibility of the digestive tract during X-ray exams. It's like a spotlight for your insides!
While barium compounds can be toxic, barium sulfate is insoluble and therefore relatively safe for this purpose.
Radium: The Radioactive Rebel
Finally, there's Radium. Radium is famous (or perhaps infamous) for its radioactivity.
Marie Curie discovered radium and its incredible properties at the turn of the 20th century. It was once used in watch dials to make them glow in the dark.
However, due to its harmful radiation, that practice has been discontinued. Radium is a reminder that even the most fascinating elements can have a dark side.
Alkaline Earth Metals: The Reactive Romantics
What makes these elements tick? Their reactivity, of course! They are all dying to get rid of those two outermost electrons.
This eagerness to react explains why you almost never find them in their pure, elemental form in nature. They're too busy bonding with other elements!
They typically form compounds with elements like oxygen and chlorine. They make compounds like magnesium oxide (MgO) and calcium chloride (CaCl2).
Everyday Encounters with Alkaline Earth Metals
These elements might seem exotic, but they're all around us! Think about it: you're practically swimming in them.
From the food you eat to the buildings you live in, alkaline earth metals play a surprisingly important role in our daily lives.
So next time you see a firework display or drink a glass of milk, remember the amazing alkaline earth metals that make it all possible. They are the unsung heroes of the periodic table, quietly supporting our world in countless ways.
The Alkaline Earth Metals and Water
The alkaline earth metals react with water, but not as violently as the group 1 elements. The reaction with water produces hydrogen gas and metal hydroxides.
The reaction gets more vigorous as you go down the group. Magnesium reacts slowly with cold water, while calcium reacts more readily.

Barium reacts quite vigorously with water. The general equation for the reaction is: M(s) + 2H2O(l) → M(OH)2(aq) + H2(g), where M is an alkaline earth metal.
Applications Beyond the Obvious
Beyond the well-known uses, alkaline earth metals find applications in various niche areas. Beryllium is used in X-ray windows because of its low absorption of X-rays.
Magnesium alloys are used in aircraft and automotive parts due to their lightweight and high strength. Calcium is used in the production of steel and other metals.
Strontium is used in some specialized glasses and ceramics. Barium is used in the production of rubber and plastics.
In Conclusion: A Toast to the Alkaline Earth Metals!
The Alkaline Earth Metals – a fascinating group of elements with a wide range of properties and applications. From the fireworks that light up our skies to the bones that support our bodies, these elements play a vital role in our world.
They are not as flashy as some other elements, but they are essential for life and technology. So, let's raise a glass (of milk, perhaps?) to the Alkaline Earth Metals – the unsung heroes of the periodic table!
They are pretty neat, right?
