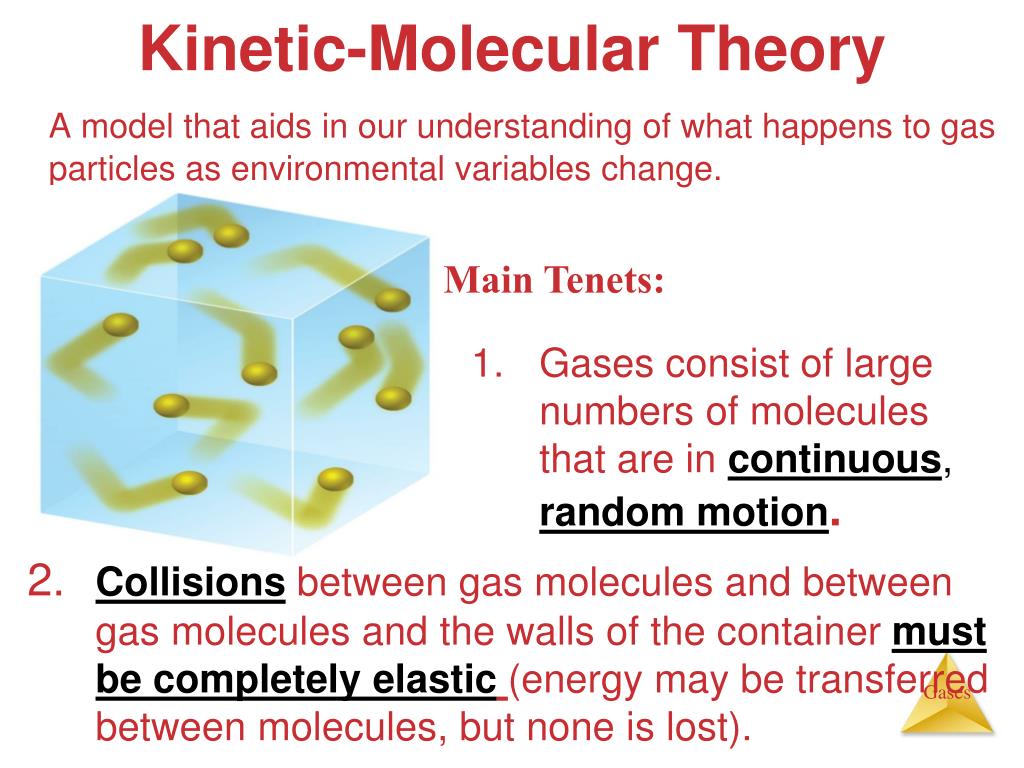
According To The Kinetic Molecular Theory

Ever wonder what's really going on inside that glass of iced tea? Or why your balloon deflates slowly over time? Well, buckle up! We're about to dive into the wild and wonderful world of the Kinetic Molecular Theory. It's not as scary as it sounds, promise!
Think of the world around you. Everything – your desk, your pet hamster, the very air you breathe – is made of tiny, tiny particles called molecules. These aren't just sitting still, minding their own business. Nope! They're bouncing around like hyperactive kids at a birthday party.
The Party Animals of the Universe
The Kinetic Molecular Theory basically says this: all matter is made of these ridiculously small particles that are constantly in motion. And the speed of this motion? It's directly related to temperature. The hotter it is, the faster they zoom! Imagine a room full of dancers. Low temperature? Slow waltz. High temperature? Full-on rave!
Must Read
It’s kind of mind-blowing, isn't it? To think that everything is just a chaotic dance of tiny particles. It’s a constant, never-ending party at a scale we can barely even imagine.
States of Mayhem (aka States of Matter)
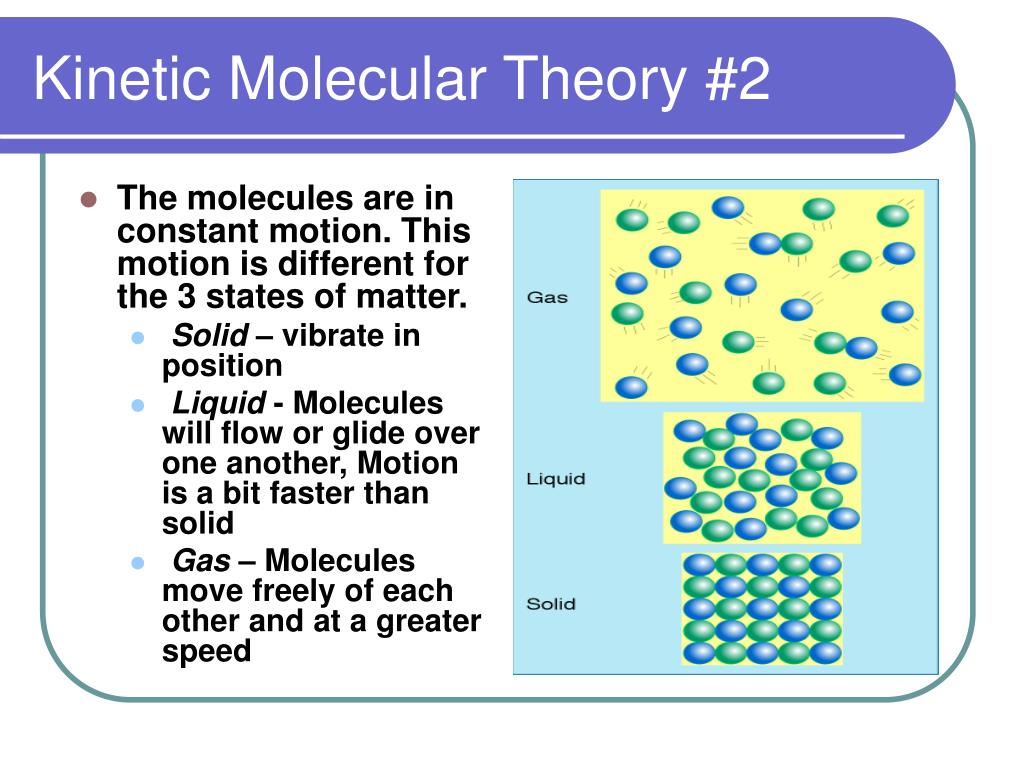
This theory also explains why we have solids, liquids, and gases. It's all about how close these particles are to each other and how much energy they have.

- Solids: These particles are tightly packed and barely moving. Think of a tightly packed crowd at a concert, only vibrating slightly. They hold their shape and volume.
- Liquids: More energy! Particles are still close together, but they can slide past each other. Like people at a slightly less crowded concert, still bumping into each other, but able to move around a bit. They hold their volume, but not their shape.
- Gases: Total anarchy! Particles are far apart and moving at breakneck speeds. Imagine that concert crowd suddenly scattered across a huge field, running in every direction. Gases have neither a fixed volume nor a fixed shape.
See? It's all about the energy and the proximity of these crazy little molecules!
Why Is This So Entertaining?
What makes the Kinetic Molecular Theory so fascinating is its power to explain so much of what we observe around us. Why does steam fog up a mirror? Because hot water molecules are flying into the air and colliding with the cooler surface. Why does sugar dissolve faster in hot coffee? Because the hot water molecules are bashing into the sugar crystals, breaking them apart faster. It's like having a secret decoder ring for the universe!
.PNG)
And the best part? It's not just some dry, academic concept. It’s a framework that helps us understand everything from the pressure in our car tires to the way our food cooks. It's a lens through which we can see the world in a completely new and exciting way.
Beyond the Basics: Real-World Fun
Think about the implications! Knowing that molecules are constantly in motion helped Robert Brown, a botanist, observe the random movement of particles suspended in a fluid. This is known as Brownian motion and it's direct evidence supporting the theory!
Or consider diffusion: that's the spreading out of particles from an area of high concentration to an area of low concentration. It explains why smells travel, why food coloring spreads in water, and a whole lot more!
"The important thing is never to stop questioning." - Albert Einstein (probably thinking about the Kinetic Molecular Theory).
So, the next time you're making a cup of tea or watching ice melt, remember the Kinetic Molecular Theory. It’s a reminder that even the most seemingly mundane things are actually governed by a hidden world of incredibly energetic, unbelievably small particles. It's a world of constant motion, collision, and interaction. A world that is, quite simply, amazing!
Ready to explore this microscopic mayhem further? Dive deeper into the physics and chemistry behind it. You might just find yourself looking at the world in a whole new way!