A Metal With 3 Valence Electrons Used In Cans
Hey, wanna talk about…metal?
Yeah, yeah, I know what you’re thinking. Boring! But trust me on this one. We’re diving into a metal that’s probably chilling in your pantry right now. The unsung hero of snacks. Ready?
Let's talk about aluminum!
Must Read
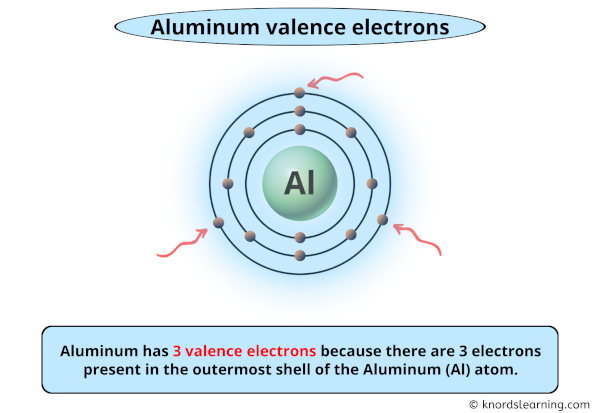
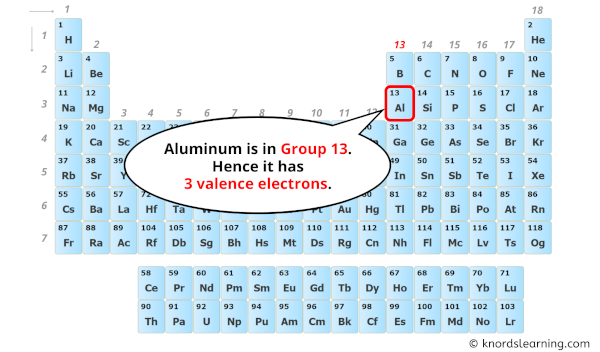
The 3-Valence Electron Superstar
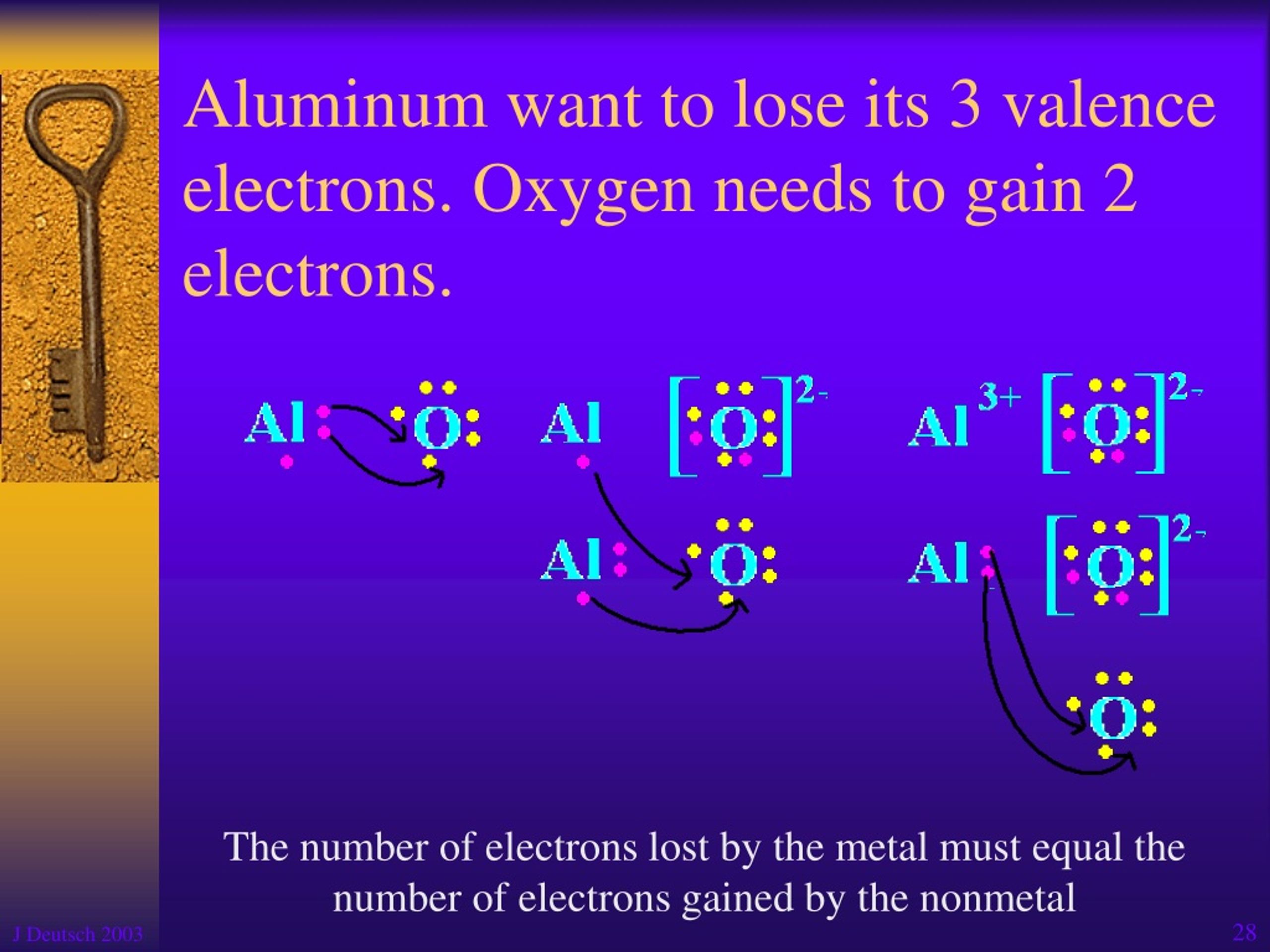
Okay, let’s get a little science-y, but I promise it won't hurt. Aluminum has three valence electrons. What does that even MEAN? Basically, it's all about how aluminum plays with other elements. These three electrons are itching to bond. It's like they’re tiny social butterflies desperate to mingle.
This electron-sharing thing is what makes aluminum so reactive, but don't worry! It's not going to spontaneously combust. Aluminum forms a super thin, but super strong, protective layer of aluminum oxide when it's exposed to air. That's what keeps it from corroding away entirely. Imagine it as a superhero shield, always protecting its metallic goodness. Pretty neat, huh?
So, three valence electrons. Remember that for your next trivia night. You'll be a star.
Cans, Cans, Everywhere!
Alright, the real reason we’re even chatting about this: aluminum cans! Look around you. Soda cans, soup cans, even some fancy sparkling water cans are often made of aluminum. Why? Loads of reasons.
First, aluminum is light. Seriously light. Imagine lugging around cans made of, say, lead. Your arms would hate you. Aluminum keeps things manageable.
Second, it’s strong. Remember that protective oxide layer we talked about? That helps keep your soda inside the can and the nasties outside the can. Nobody wants rusty soda.
Third, and this is a big one: aluminum is recyclable! Like, really recyclable. You can melt it down and turn it into something new over and over and over again. Talk about a sustainable superstar! So toss those cans in the blue bin, knowing you're helping the planet.
Think about it: every time you crack open a can of your favorite beverage, you're interacting with a metal powered by those three little valence electrons. It's kinda mind-blowing when you think about it, right?
Quirky Aluminum Facts (Because Why Not?)
Okay, time for some random aluminum trivia to impress your friends (or just yourself).
Did you know that aluminum is the most abundant metal in the Earth's crust? It’s practically everywhere. Under your feet, in mountains, all over the place. It's just a matter of getting it out in its pure form.

And here’s a weird one: Aluminum foil isn’t actually 100% pure aluminum. It's usually an alloy (a mix of metals) with a tiny bit of other stuff added to make it stronger. The more you know!
Another fun fact: Aluminum is used in space travel! It's lightweight, strong, and can withstand extreme temperatures. So, yeah, aluminum is literally out of this world.
Why Should You Care? (Besides the Trivia Night Glory)
So, why bother learning about aluminum and its three valence electrons?
Well, for starters, it makes you a more informed consumer. You can appreciate the engineering and science that goes into everyday objects, like your humble soda can.

Plus, understanding the properties of materials helps you make smarter choices. Recycling aluminum saves energy and resources. By being aware of the benefits of aluminum, you can contribute to a more sustainable future.
And, honestly, it's just cool to know stuff! Knowing that a seemingly simple can of soda is a testament to materials science and responsible manufacturing is pretty awesome.
Aluminum: Not Just a Dull Can
So, there you have it: a whirlwind tour of aluminum, its three valence electrons, and its role in the world of cans (and beyond!). It's more than just a shiny metal; it's a versatile, recyclable, and frankly, fascinating material that touches our lives every day.
Next time you reach for a can, take a moment to appreciate the aluminum and its tiny, but mighty, electrons. You might just see the world in a whole new (metallic) light! Go forth and spread the aluminum love!
