6p Angular Momentum Quantum Number

Hey there, science enthusiast! Ever hear someone toss around the term "6p" and feel like you've stumbled into a secret agent convention? Don't worry, it's not nearly as cloak-and-dagger as it sounds. Today, we're diving into the wonderfully weird world of quantum mechanics to decode the mysterious 6p Angular Momentum Quantum Number. Buckle up, it's gonna be a (relatively) smooth ride!
First things first, what is angular momentum? Imagine a tiny electron zipping around the nucleus of an atom. It's not just going around in a simple circle (sorry, Bohr model, you tried!). It’s moving in a more complex, three-dimensional way, creating what we call angular momentum. Think of it like a figure skater doing a spin – the faster they spin, the more angular momentum they have.
Now, quantum mechanics gets involved, and things get a bit… quantized. That means angular momentum can't just be any value; it can only exist in specific, discrete amounts. That's where our friend, the angular momentum quantum number (l), comes in. It tells us the shape of the electron's orbital – where it's most likely to be hanging out. Think of it like the electron’s preferred dance moves!
Must Read
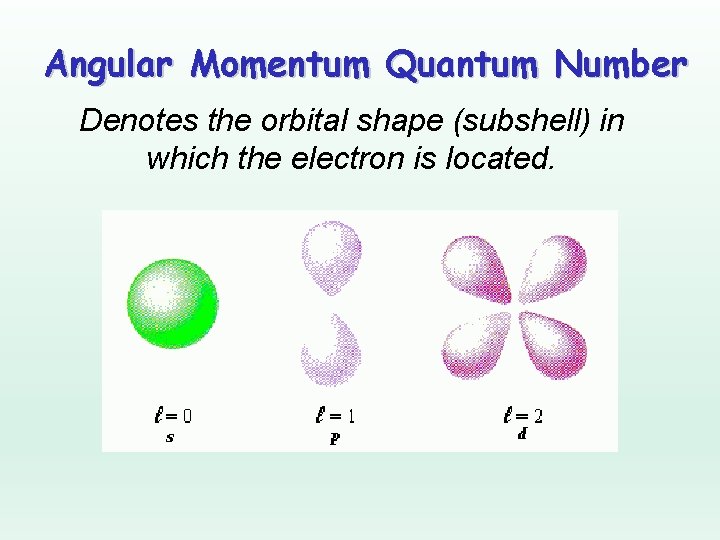
The values of 'l' start at 0 and can go up to n-1, where 'n' is the principal quantum number (1, 2, 3, etc. - think of it as the energy level). Each value of 'l' is assigned a letter:
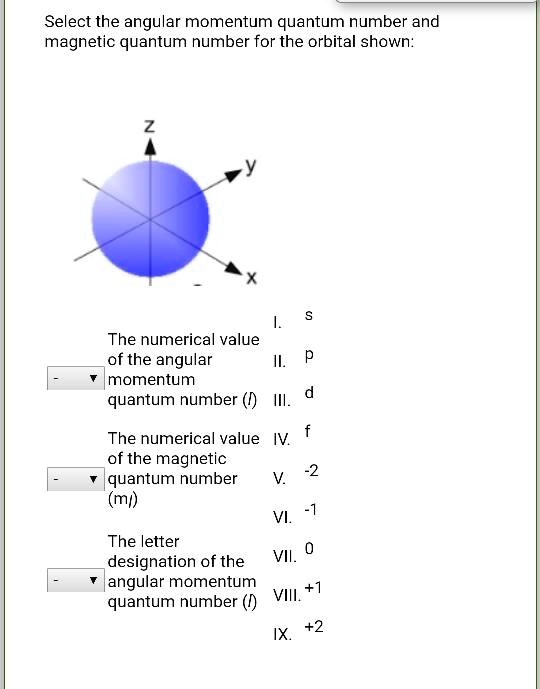
- l = 0 is the s orbital (spherical, like a perfectly round soccer ball)
- l = 1 is the p orbital (dumbbell-shaped, like a peanut in a shell, but with two lobes)
- l = 2 is the d orbital (more complex shapes, some looking like a four-leaf clover)
- l = 3 is the f orbital (even more complex – seriously, Google it; they're kinda wild!)
And so on... but let's stick with 'p' for now!

Decoding the "6p"
Okay, so what does "6p" actually mean? It’s pretty simple once you break it down. The '6' refers to the principal quantum number (n). This tells us the energy level – the electron's general distance from the nucleus. A '6' means it's in the 6th energy level, further out than electrons in the 1st or 2nd levels.
The 'p' tells us the shape of the orbital, thanks to our trusty angular momentum quantum number (l=1). So, a 6p electron is buzzing around in the 6th energy level, primarily hanging out in a dumbbell-shaped p orbital.
Think of it like this: the number is the floor of an apartment building, and the letter is the shape of the apartment. A 6p electron lives in the 6th floor in a "p-shaped" apartment. Makes sense, right? (I hope so, otherwise, I've been using this analogy wrong for years!)
But wait, there's more! Each p orbital has three possible orientations in space. Remember how the p orbital looks like a dumbbell? Well, that dumbbell can point along the x-axis (px), the y-axis (py), or the z-axis (pz). These are called the magnetic quantum numbers (ml), which range from -l to +l (including 0). So, for p orbitals (l=1), ml can be -1, 0, or +1, corresponding to px, py, and pz, respectively.
Therefore, a 6p subshell can hold up to 6 electrons in total - two in each of the three p orbitals (px, py, and pz).
Why Does Any of This Matter?
Great question! Understanding electron configurations (like knowing we have 6p electrons) is crucial for predicting how atoms will interact with each other to form molecules and chemical bonds. It helps us understand why some elements are reactive and others are inert, why some substances are colorful and others are not. In other words, it’s the bedrock of chemistry!

Plus, thinking about these tiny, quantized particles can be a little mind-bending, right? It reminds us that the universe is far stranger and more fascinating than we can easily imagine.
So, the next time you hear "6p," you can confidently nod and say, "Ah yes, an electron in the 6th energy level, residing in a dumbbell-shaped orbital!" You'll sound like a quantum physics wizard (or at least someone who read a really cool article online).
Congratulations, you've just leveled up your science knowledge! Isn't it amazing how much there is to learn about the universe? Keep exploring, keep questioning, and never stop being curious. The world of science is waiting for you, one quantum leap at a time!
