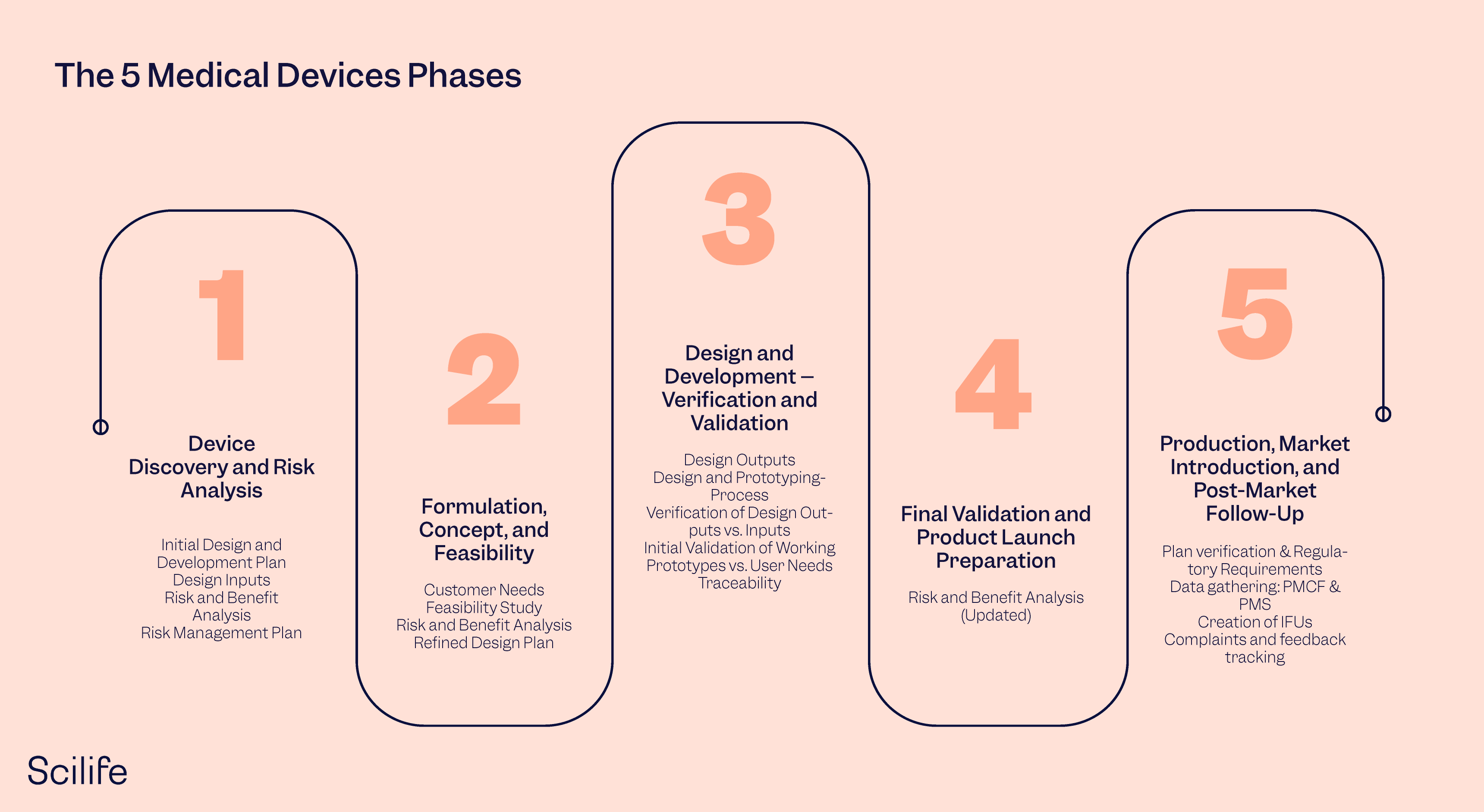
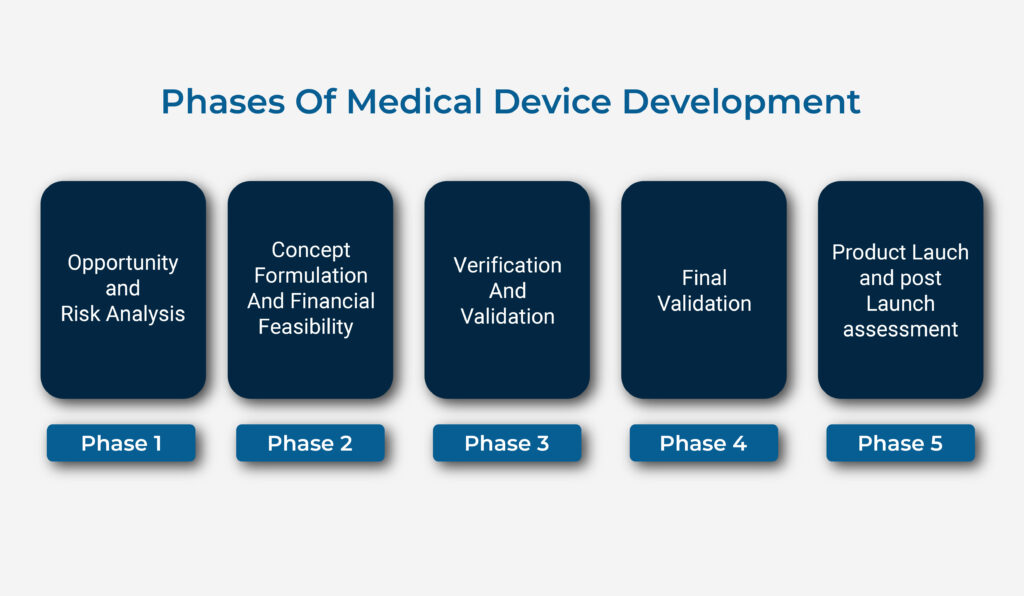
5 Phases Of Medical Device Development
Ever wondered how that amazing gizmo in your doctor's office – you know, the one that goes "beep boop" and makes you feel like you're in a sci-fi movie – actually gets there? It's not magic! Well, okay, maybe a little magic, but mostly it's a whole lotta science, engineering, and a sprinkle of regulatory paperwork.
Developing a medical device is like baking a cake, only instead of a cake, you're making something that might save someone's life. And instead of flour and sugar, you're using circuits, software, and maybe even some lasers! It’s a process!
Phase 1: The Eureka! Moment (or: "I have an Idea!")
Every great invention starts with an idea. Maybe you've got a brilliant thought while sipping coffee. Or maybe while you are thinking about making a new toaster...
Must Read
This is the “Eureka!” phase, the “lightbulb” moment, the “I’ve got it!” epiphany. This is where the idea, the seed, for your amazing new medical device is born. Imagine a doctor saying, "Hey, I wish there was a way to..." and BOOM! An idea is born!
Let's say, for example, you dream up a sock that monitors blood sugar. This sock sings a little tune when your blood sugar is low. (Okay, maybe not the singing part, but you get the idea!). This sock can change the world!
This phase is all about dreaming big and scribbling ideas on napkins. Lots of napkins! Don't worry about the details yet. Just let the creativity flow. Think of it like brainstorming with your brain!
Is my idea even possible?
You should research if it's even possible. It's time to see if it's already been invented. (Because, let's be honest, someone probably thought of it before you!).
Patent databases and scientific literature become your new best friends. Don’t be discouraged if someone else has a similar idea. See how you can improve upon it. Maybe you can make it smaller, faster, or… sing better!
Phase 2: Proof of Concept (Can we actually build this thing?)
Now that you have a great idea, it’s time to see if it’s even remotely possible. Can you turn your napkin scribbles into something real? It is like trying to build a rocket in your garage.
![Medical Device Development Process [5 Phases]](https://blog.greenlight.guru/hubfs/5 Phase Development of a New Medical Device.png)
This is where you build a prototype. A clunky, messy, duct-tape-and-glue version of your device. This is your “proof of concept,” your “Frankenstein’s monster,” your way of saying, "See? I can almost do it!"
Let's go back to our singing blood sugar sock. The prototype might involve a regular sock, some wires, a blood sugar monitor taped to it, and a tiny speaker. It might look ridiculous, but if it sort of works, you’re on the right track!
This phase is all about experimentation. Trial and error. Lots and lots of error! You will likely face many roadblocks!
Funding this crazy thing
You need money to create things! This can involve pitching your idea to investors, applying for grants, or maxing out your credit cards (maybe don't do that last one!).
Think of it as your own personal "Shark Tank." You have to convince people that your idea is worth investing in. Even if your prototype looks like it was made by a five-year-old with a glue gun.
Phase 3: Design and Development (Making it pretty… and functional!)
Alright, you have proven that your idea is possible. You even got some funding. Now it’s time to turn that clunky prototype into a sleek, functional, and manufacturable device.

This phase is where the real engineering magic happens. This involves teams of engineers, designers, and regulatory experts. They work together to refine the design, optimize the performance, and ensure the device meets all the necessary safety and regulatory requirements.
Our singing blood sugar sock needs a serious makeover. We're talking miniaturized sensors, comfortable fabric, a discreet speaker, and maybe even a Bluetooth connection to your phone. It needs to be something people would actually want to wear!
This phase is iterative. Lots of testing, tweaking, and re-testing. Think of it as a never-ending cycle of improvement. Until you get it perfect!
Dealing with the Regulators
Medical devices are heavily regulated to ensure patient safety. You'll need to navigate a maze of regulations, standards, and guidelines.
Organizations like the FDA (in the United States) and similar agencies in other countries will scrutinize your device. They want to make sure it’s safe and effective before it hits the market. This is the “adulting” part of the process, where you need to dot your "i"s and cross your "t"s.
Phase 4: Clinical Trials (Does it actually work… on humans?)
So, you’ve built a beautiful, functional device. It looks great on paper. But does it actually work in the real world? On real people?
![Medical Device Development Process [5 Phases]](https://www.greenlight.guru/hs-fs/hubfs/Understanding the 5 Phases of Medical Device Development-1.png?width=1600&height=800&name=Understanding the 5 Phases of Medical Device Development-1.png)
This is where clinical trials come in. These are studies where you test your device on human subjects. This determines whether it’s safe and effective. It’s like putting your device through its paces in a real-world scenario.
We need to test our singing blood sugar sock on a group of people with diabetes. We need to see if it accurately monitors their blood sugar and alerts them when it’s low. And, of course, if they find the singing annoying!
Clinical trials can be long, expensive, and stressful. You're at the mercy of the data. But they're essential for proving that your device is safe and effective.
Analyzing the Data
After the clinical trials, you analyze the data. Then see if your device actually does what it’s supposed to do. Statisticians become your new best friends.
If the data looks good, you can move on to the next phase. If not, it’s back to the drawing board. It can be disappointing but it's also an opportunity to improve.
Phase 5: Manufacturing and Launch (Behold! The finished product!)
Hallelujah! Your device has passed all the tests. It’s been approved by the regulators. Now it’s time to mass-produce it and unleash it upon the world!
This phase involves setting up manufacturing facilities, training staff, and developing a marketing strategy. It's also time to get your device into the hands of the people who need it.
Our singing blood sugar sock is finally ready to hit the market! We need to manufacture millions of them. We also need to get them into pharmacies, hospitals, and online stores.
This is the most exciting phase. Seeing your creation helping people and improving their lives. It's a moment of pure satisfaction.
Post-Market Surveillance
The journey doesn’t end with the launch. You need to continue monitoring your device. Even after it’s on the market.
This is called post-market surveillance. It involves tracking any adverse events. Reporting issues, and making improvements to the device as needed. This ensures the device remains safe and effective throughout its lifespan.
So there you have it! The five phases of medical device development. It’s a long, challenging, and expensive process, but it’s also incredibly rewarding.
You are creating something that can make a real difference in the world. Who knows? Maybe you will invent the next life-saving medical device! Now, who's up for inventing a teleporter?
